��Ŀ����
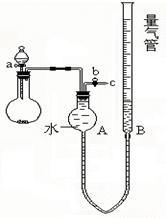
(9��)A��B��C����ѧ���������ַǽ������ʣ�������BΪ��ɫ���壬A��CΪ���壬�ס��ҡ���Ϊ��ѧ���������ֻ����������C���������У�A��B��C��ס��ҡ���֮���ת����ϵ����ͼ��ʾ��
(1)д��Cת��Ϊ���Ļ�ѧ����ʽ________________________________________��
(2)�����£�����ͼװ��װ�ã�����������ѹǿ����ʱ��ˮ����Һ����M����ȡ����Ƥ�����ڲ���ȼ�ճ��м����ң�����B�г��ȼ�գ�ˮ����������ܡ�
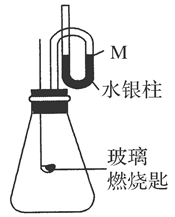
��Ҫ�ش��������⣺
��д����ȼ�յĻ�ѧ����ʽ��_____________________________________________��
��ˮ����������ܵ�ԭ����______________________________________________��
�ۻ���Ϩ�����һ��ʱ��ָ���ԭ�¶ȣ��ҹ�ˮ����λ��Ӧ����________��
A������M��������B������M��������C������M��
����ѡ���Ҫ˵������_______________________________________________��
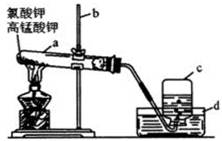
������ͼ��ʾװ�ü�����ȼ�յIJ���A��B��C��D����ʢ�ŵ�ҩƷ�����ǣ�
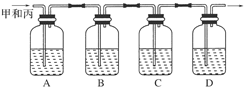
A��________��B��________��C��________��D��________

(1)д��Cת��Ϊ���Ļ�ѧ����ʽ________________________________________��
(2)�����£�����ͼװ��װ�ã�����������ѹǿ����ʱ��ˮ����Һ����M����ȡ����Ƥ�����ڲ���ȼ�ճ��м����ң�����B�г��ȼ�գ�ˮ����������ܡ�

��Ҫ�ش��������⣺
��д����ȼ�յĻ�ѧ����ʽ��_____________________________________________��
��ˮ����������ܵ�ԭ����______________________________________________��
�ۻ���Ϩ�����һ��ʱ��ָ���ԭ�¶ȣ��ҹ�ˮ����λ��Ӧ����________��
A������M��������B������M��������C������M��
����ѡ���Ҫ˵������_______________________________________________��
������ͼ��ʾװ�ü�����ȼ�յIJ���A��B��C��D����ʢ�ŵ�ҩƷ�����ǣ�

A��________��B��________��C��________��D��________
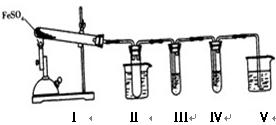
(1)S��O2SO2 (2)��CS2��3O2CO2��2SO2 ��CS2ȼ���Ƿ��ȷ�Ӧ���¶����ߣ�ʹ�����������ѹǿ����A��CS2ȼ��ǰ����������ʵ����ޱ仯 ��Ʒ����Һ�����Ը��������Һ��Ʒ����Һ������ʯ��ˮ
��

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
 ______������ţ�����ȱ�ٵ������� ��
______������ţ�����ȱ�ٵ������� ��

 ����������̽����ʵ���������ɷ֡������ʵ����ƣ���д�����Լ���Ԥ����������ۡ���ѡ�Լ���3mol��L��1H2SO4��6 mol��L��1NaOH��0.5 mol��L��1BaCl2��0.5 mol��
����������̽����ʵ���������ɷ֡������ʵ����ƣ���д�����Լ���Ԥ����������ۡ���ѡ�Լ���3mol��L��1H2SO4��6 mol��L��1NaOH��0.5 mol��L��1BaCl2��0.5 mol�� L��1Ba(NO3)2��0.01 mol��L��1����KMnO4��Һ��0.01 mol��L��1��ˮ
L��1Ba(NO3)2��0.01 mol��L��1����KMnO4��Һ��0.01 mol��L��1��ˮ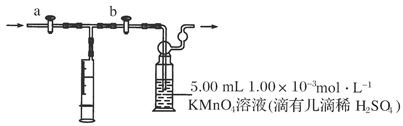
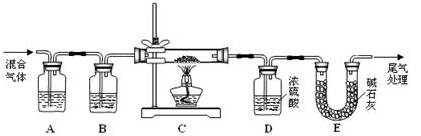
 CO�� + CO2�� + H2O��
CO�� + CO2�� + H2O��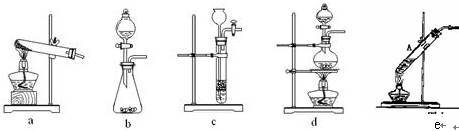
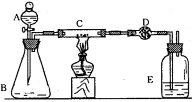
 װ�ÿ�����_____________������ʢ�ŵ�ҩƷ��____________��
װ�ÿ�����_____________������ʢ�ŵ�ҩƷ��____________��