��Ŀ����
���ǵ����Ϻ����ḻ��һ��Ԫ�أ������仯�����ڹ�ũҵ������������������Ҫ���á���ش��������⣺
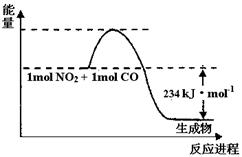
��1����ͼ1��1 mol NO2��1mol CO��Ӧ����CO2��NO�����������仯ʾ��ͼ��ͼ���漰���ʾ�Ϊ��̬������д��NO2��CO��Ӧ���Ȼ�ѧ����ʽ����������
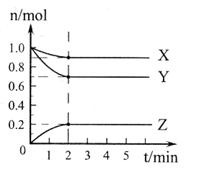
��2����0.5L���ܱ������У�����һ�����ĵ������������з�Ӧ��������������ʵ�����ʱ��ı仯������ͼ2��ʾ����Y������������2min��N2�ķ�Ӧ���ʦ�(N2)=����������
 ������
������
����ͼ1������������������������������������ͼ2
��3�����Ṥҵ�ڹ��á�������ҵ�ͺ�����ҵ��ռ����Ҫ��λ�����Ż�ѧ��ά�������л��ϳɡ�����������ȹ�ҵ��Ѹ�ٷ�չ����Ҫʹ�ô��������ᡣ
�ٹ�ҵ�������ò�ϵ������Ϊ�������ð���������ȡ���ᡣ��һ����ӦΪ���ڴ���������������������Ӧ����NO��д����Ӧ��ѧ����ʽ����������
��ͭ������ϡ���ᷴӦ��Ҳ����Ũ���ᷴӦ����ͭ��һ��Ũ�����ᷴӦʱ���ɽ�����ʽ��ʾΪ��Cu+HNO3 ��Cu(NO3)2+NO��+NO2��+H2O ������ʽδ��ƽ������Ӧ�л�ԭ����������������0.6mol Cu��������ȫ�ܽ������õ���NO��NO2���ʵ�����ͬ����õ���״�����������Ϊ��������L��
��1����ͼ1��1 mol NO2��1mol CO��Ӧ����CO2��NO�����������仯ʾ��ͼ��ͼ���漰���ʾ�Ϊ��̬������д��NO2��CO��Ӧ���Ȼ�ѧ����ʽ����������
��2����0.5L���ܱ������У�����һ�����ĵ������������з�Ӧ��������������ʵ�����ʱ��ı仯������ͼ2��ʾ����Y������������2min��N2�ķ�Ӧ���ʦ�(N2)=����������
 ������
������
����ͼ1������������������������������������ͼ2
��3�����Ṥҵ�ڹ��á�������ҵ�ͺ�����ҵ��ռ����Ҫ��λ�����Ż�ѧ��ά�������л��ϳɡ�����������ȹ�ҵ��Ѹ�ٷ�չ����Ҫʹ�ô��������ᡣ
�ٹ�ҵ�������ò�ϵ������Ϊ�������ð���������ȡ���ᡣ��һ����ӦΪ���ڴ���������������������Ӧ����NO��д����Ӧ��ѧ����ʽ����������
��ͭ������ϡ���ᷴӦ��Ҳ����Ũ���ᷴӦ����ͭ��һ��Ũ�����ᷴӦʱ���ɽ�����ʽ��ʾΪ��Cu+HNO3 ��Cu(NO3)2+NO��+NO2��+H2O ������ʽδ��ƽ������Ӧ�л�ԭ����������������0.6mol Cu��������ȫ�ܽ������õ���NO��NO2���ʵ�����ͬ����õ���״�����������Ϊ��������L��
��1��NO2(g) + CO(g) = CO2(g) + NO(g) ���SH =��234kJ��mol-1��2�֣�
��2��H2��1�֣���0.1mol?L-1?min-1����1�֣���д��λ�����֣���ͬ��
��3����4NH3+5O2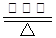 4NO+6H2O������2�֣� ��NO��NO2��1�֣���13.44 ��2�֣�
4NO+6H2O������2�֣� ��NO��NO2��1�֣���13.44 ��2�֣�
��2��H2��1�֣���0.1mol?L-1?min-1����1�֣���д��λ�����֣���ͬ��
��3����4NH3+5O2
 4NO+6H2O������2�֣� ��NO��NO2��1�֣���13.44 ��2�֣�
4NO+6H2O������2�֣� ��NO��NO2��1�֣���13.44 ��2�֣���1������ͼ1��֪����Ӧ���������������������������������Ƿ��ȷ�Ӧ���Ȼ�ѧ����ʽΪNO2(g) + CO(g) = CO2(g) + NO(g) ���SH =��234kJ��mol-1��
��2������ͼ���֪��X��Y�����ʵ�����С��ƽ��ʱ�ֱ����0.1mol��0.3mol������Y��������X�ǵ����������ķ�Ӧ������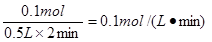
��3���ٰ��������ķ���ʽΪ4NH3+5O2 4NO+6H2O��
4NO+6H2O��
������������ԭ�����ɵ������ǻ�ԭ�����������Ļ�ԭ������NO��NO2��0.6molͭʧȥ1.2mol���ӣ����NO��NO2�����ʵ�����1.2mol��4��0.3mol�����������6.72L�������ܹ���13.44 L��
��2������ͼ���֪��X��Y�����ʵ�����С��ƽ��ʱ�ֱ����0.1mol��0.3mol������Y��������X�ǵ����������ķ�Ӧ������
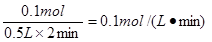
��3���ٰ��������ķ���ʽΪ4NH3+5O2
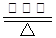 4NO+6H2O��
4NO+6H2O��������������ԭ�����ɵ������ǻ�ԭ�����������Ļ�ԭ������NO��NO2��0.6molͭʧȥ1.2mol���ӣ����NO��NO2�����ʵ�����1.2mol��4��0.3mol�����������6.72L�������ܹ���13.44 L��

��ϰ��ϵ�д�
�����Ŀ
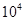 L����״������β����42��5gNH3����x= ��
L����״������β����42��5gNH3����x= ��