��Ŀ����
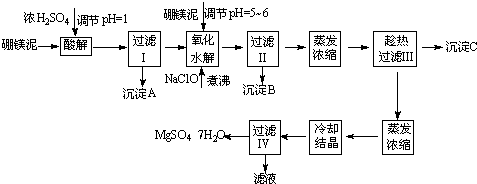
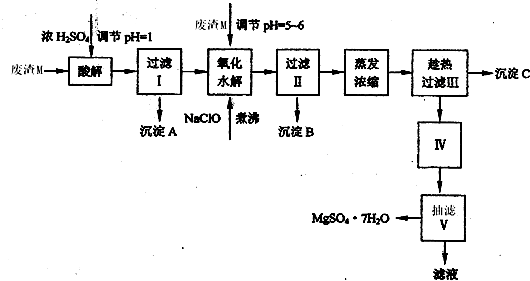
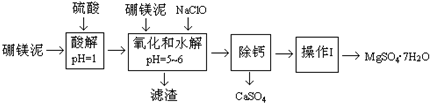
MgSO4?7H2O��������ֽ����֯���մɡ����Ṥҵ��Ҳ����ҽ��������к�Σ�ij��������Ҫ�ɷ���MgCO3�������CaCO3��SiO2�����ʣ��Ӵ˷����л���MgSO4?7H2O�Ĺ����£�
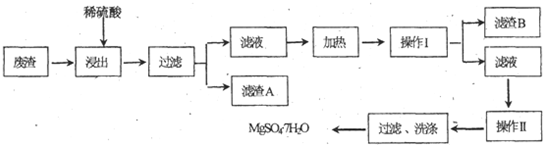
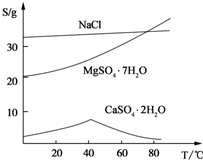
��֪CaSO4��MgSO4���ܽ�ȣ�S�����¶ȣ�T���仯������ͼ��ʾ���Իش��������⣺
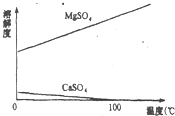
��1���������������У�Ϊ���þ�Ľ����ʣ��ɲ�ȡ�Ĵ�ʩ��______��Ҫ��д������һ������
��2������B����Ҫ�ɷֵĻ�ѧʽ��______��
��3���������������______���������������______��
��4��������Ͳ�������Ҫ�õ����������������в���������Ҫ������______���������в���������Ҫ������______��
��5��������ΪMg2+��ˮ�⣬����������ٹ��ˡ�ϴ�ӵò���MgSO4?7H2O���壬����Ϊ����˵���Ƿ���ȷ______����ǡ�����ԭ����______��
�⣺��1����ϡ�����ڷ����У�MgCO3��CaCO3�ܽ⣬�����ܽ��-�¶�ͼ��֪��MgSO4���¶����߶��ܽ�����������������У�Ϊ���þ�Ľ����ʣ��ɲ�ȡ�Ĵ�ʩ�ʵ���߷�Ӧ�¶ȣ�ͬʱ���Ͻ��裬���ӽ���ʱ�䣮
�ʴ�Ϊ���ʵ���߷�Ӧ�¶ȣ�ͬʱ���Ͻ��裬���ӽ���ʱ�䣮
��2���ɹ������̿�֪����������������ˣ���Һ�к���CaSO4��MgSO4�������ܽ��-�¶�ͼ��֪��CaSO4���¶����߶��ܽ���½���MgSO4���¶����߶��ܽ��������Һ��������CaSO4���ʲ������ڼ��ȵ�����¹��ˣ�����B��CaSO4��
�ʴ�Ϊ��CaSO4��
��3��CaSO4���¶����߶��ܽ���½������ȵ�����£����Խ���CaSO4�ܽ�ȣ��������ڼ��ȵ�����½���������Һ���룬Ӧ�dz��ȹ��ˣ�������������Һ���������壬ӦΪ����Ũ������ȴ�ᾧ��
�ʴ�Ϊ�����ȹ��ˣ�����Ũ������ȴ�ᾧ��
��4���������ǹ��ˣ�������Ϊ�������ã��������ǽᾧ��������Ϊ�������ã�
�ʴ�Ϊ�����������裮
��5��MgSO4ˮ������������þ�����ᣬ�������ѻӷ����ᣬ������ɵ�������������þ����������þ�����������ˡ�ϴ�ӿ��Եõ�MgSO4?7H2O���壮
�ʴ�Ϊ�����������ѻӷ����ᣮ
��������1����ϡ�����ڷ����У�MgCO3��CaCO3�ܽ⣬�����ܽ��-�¶�ͼ��֪��MgSO4���¶����߶��ܽ������
��2���ɹ������̿�֪����������������ˣ���Һ�к���CaSO4��MgSO4�������ܽ��-�¶�ͼ��֪��CaSO4���¶����߶��ܽ���½���MgSO4���¶����߶��ܽ��������Һ��������CaSO4��
��3���ɹ������̿�֪�����������ڼ��ȵ�����½���������Һ���룬������������Һ���������壮
��4���������ǹ��ˣ�������Ϊ�������ã��������ǽᾧ��������Ϊ�������ã�
��5��MgSO4ˮ������������þ�����ᣬ�����Ƿǻӷ����ᣮ
���������⿼��ѧ���Ķ���Ŀ��ȡ��Ϣ�������Թ������̵����⡢�����ƶϡ����ʷ����ᴿ���������Ƶȣ��Ѷ��еȣ���Ҫѧ���߱���ʵ�Ļ���֪ʶ���ۺ�����֪ʶ����Ϣ���н�������������
�ʴ�Ϊ���ʵ���߷�Ӧ�¶ȣ�ͬʱ���Ͻ��裬���ӽ���ʱ�䣮
��2���ɹ������̿�֪����������������ˣ���Һ�к���CaSO4��MgSO4�������ܽ��-�¶�ͼ��֪��CaSO4���¶����߶��ܽ���½���MgSO4���¶����߶��ܽ��������Һ��������CaSO4���ʲ������ڼ��ȵ�����¹��ˣ�����B��CaSO4��
�ʴ�Ϊ��CaSO4��
��3��CaSO4���¶����߶��ܽ���½������ȵ�����£����Խ���CaSO4�ܽ�ȣ��������ڼ��ȵ�����½���������Һ���룬Ӧ�dz��ȹ��ˣ�������������Һ���������壬ӦΪ����Ũ������ȴ�ᾧ��
�ʴ�Ϊ�����ȹ��ˣ�����Ũ������ȴ�ᾧ��
��4���������ǹ��ˣ�������Ϊ�������ã��������ǽᾧ��������Ϊ�������ã�
�ʴ�Ϊ�����������裮
��5��MgSO4ˮ������������þ�����ᣬ�������ѻӷ����ᣬ������ɵ�������������þ����������þ�����������ˡ�ϴ�ӿ��Եõ�MgSO4?7H2O���壮
�ʴ�Ϊ�����������ѻӷ����ᣮ
��������1����ϡ�����ڷ����У�MgCO3��CaCO3�ܽ⣬�����ܽ��-�¶�ͼ��֪��MgSO4���¶����߶��ܽ������
��2���ɹ������̿�֪����������������ˣ���Һ�к���CaSO4��MgSO4�������ܽ��-�¶�ͼ��֪��CaSO4���¶����߶��ܽ���½���MgSO4���¶����߶��ܽ��������Һ��������CaSO4��
��3���ɹ������̿�֪�����������ڼ��ȵ�����½���������Һ���룬������������Һ���������壮
��4���������ǹ��ˣ�������Ϊ�������ã��������ǽᾧ��������Ϊ�������ã�
��5��MgSO4ˮ������������þ�����ᣬ�����Ƿǻӷ����ᣮ
���������⿼��ѧ���Ķ���Ŀ��ȡ��Ϣ�������Թ������̵����⡢�����ƶϡ����ʷ����ᴿ���������Ƶȣ��Ѷ��еȣ���Ҫѧ���߱���ʵ�Ļ���֪ʶ���ۺ�����֪ʶ����Ϣ���н�������������

��ϰ��ϵ�д�
�����Ŀ