��Ŀ����
14��ԭ���������������X��Y��Z��M��WΪ���ֶ�����Ԫ�أ���֪Y��Z��M��W�ֱ�ͬ���ڣ�X��Mͬ���壻X��Z��M��������֮�͵���Y��W��������֮�ͣ�X��Z��M��Z�����Էֱ��γ�A2B��A2B2�͵Ļ����X��Y�γɵ���̬�������ڱ�״���µ���Է�������Ϊ17��Y��M��W������������ˮ��������䶼�ܷ�����Ӧ������˵����ȷ���ǣ�������| A�� | M�ĵ��ʿ����ڴ��ѡ�ﯵȽ������Ȼ�����Һ���û����ѡ�ﯵȵ��� | |
| B�� | ��X��Y��Z����Ԫ���γɵ�ij�ֻ������ˮ��Һ�����ܳʼ��Ի����� | |
| C�� | ��X��M��Z�γɵĻ�����X2Z2��M2Z2�������Ļ�ѧ��������ͬ | |
| D�� | ����W�����ӵ�ij���Σ���������ˮ�� |
���� ԭ���������������X��Y��Z��M��WΪ���ֶ�����Ԫ�أ�X��Y�γɵ���̬���������Է�������Ϊ17��������ӦΪNH3������֪XΪHԪ�ء�YΪNԪ�أ�X��Mͬ���壬Mԭ����������NԪ�أ���MΪNa��Y��Zͬ���ڣ�X��Z��M��Z�����Էֱ��γ�A2B��A2B2�͵Ļ������ZΪOԪ�أ�M��Wͬ���ڣ�X��Z��M��������֮�͵���Y��W��������֮�ͣ���M��Wԭ���������1+8-7=2������֪WΪAlԪ�أ�Y��M��W������������ˮ��������䶼�ܷ�����Ӧ���������⣬�ݴ˽��
��� �⣺ԭ���������������X��Y��Z��M��WΪ���ֶ�����Ԫ�أ�X��Y�γɵ���̬���������Է�������Ϊ17��������ӦΪNH3������֪XΪHԪ�ء�YΪNԪ�أ�X��Mͬ���壬Mԭ����������NԪ�أ���MΪNa��Y��Zͬ���ڣ�X��Z��M��Z�����Էֱ��γ�A2B��A2B2�͵Ļ������ZΪOԪ�أ�M��Wͬ���ڣ�X��Z��M��������֮�͵���Y��W��������֮�ͣ���M��Wԭ���������1+8-7=2������֪WΪAlԪ�أ�Y��M��W������������ˮ��������䶼�ܷ�����Ӧ���������⣮
A��Na�Ļ�ԭ�Ժ�ǿ���ܴ��ѡ�ﯵȽ������Ȼ������û����ѡ�ﯵȵ��ʣ������ܴ����ǵ��Ȼ�����Һ���û������ʣ�����Na��ˮ��Ӧ����������������������A����
B����H��N��O����Ԫ���γɵ�ij�ֻ����������Һ�����ԣ���һˮ�ϰ���ҺΪ���ԣ���B��ȷ��
C��H2O2��ֻ���й��ۼ�����Na2O2�к��й��ۼ������Ӽ��������Ļ�ѧ�����Ͳ���ͬ����C����
D������Al3+���ӵ���--��������������ˮ������D��ȷ��
��ѡBD��
���� ���⿼��ṹ����λ�ù�ϵӦ�ã��ƶ�Ԫ���ǽ���ؼ���ע��Aѡ��Ϊ�״��㣮

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д����ܱ������н�����CuBr2��487K�¼��ȷֽ⣬ƽ��ʱp��Br2��Ϊ4.66��103Pa��
�練Ӧ�¶Ȳ��䣬����Ӧ��ϵ���������һ������p��Br2���ı仯��ΧΪ��������
| A�� | p��Br2����4.66��103Pa | B�� | 2.33��103Pa��p��Br2����4.66��103Pa | ||
| C�� | ��p��Br2����2.33��103Pa | D�� | 2.33��103Pa��p��Br2����4.66��103Pa |
| A�� | ����������������ɢϵ�ı��������Ƿ�ɢ�ʵ���ֱ����10-9��10-7m֮�� | |
| B�� | һ������������ʱ�������пɷ��������ЧӦ | |
| C�� | ����ķ�ɢ������ֱ������Һ�Ĵʽ�����������ֽ | |
| D�� | Fe��OH��3�����ܹ�ʹˮ�������Ĺ�������������ﵽ��ˮĿ�� |
| A�� | Ũ�� | B�� | �¶� | C�� | ѹǿ | D�� | ���� |
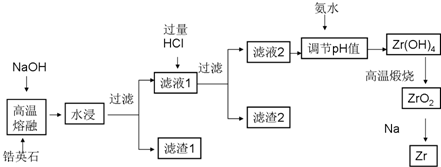
��֪25��ʱ����������������������ʽ��ȫ����ʱ��Һ��pH���±���
| ������ | Zr��OH��4 | Fe��OH��3 | Al��OH��3 | Fe��OH��2 |
| ��ʼ����ʱpH | 1.2 | 2.7 | 3.4 | 6.3 |
| ��ȫ����ʱpH | 2.4 | 3.2 | 5.2 | 9.7 |
ZrSiO4+4NaOH=Na2ZrO3+Na2SiO3+2H2O
��2��Ϊ����߽����ʿɲ�ȡ�ķ����н�ԭ�Ϸ�������Ӵ���������Ȼ������ν�ȡ���о����֣�
��3����������2�ijɷ�H2SiO3 �� H4SiO4���û�ѧʽ��ʾ��
��4���ð�ˮ����Һ2��pH����2.4��pH��3.4��Ŀ����ʹZr4+��ȫת��ΪZr��OH��4������ͬʱ��֤Al3+���ᱻ����������
��5��������������1�м���������Һ�����˲�������ٽ���Һ�ڲ���ͨ��HCl����������ͬʱ�����ᾧ���ɻ��յõ������Ȼ���FeCl3���壮

��֪��
���������ᷴӦ�Ļ�ѧ����ʽΪ��Co+2HCl�TCoCl2+H2��
��CoCl2•6H2O�۵�86�棬������ˮ�����ѵȣ��������ȶ�����������110��120��ʱ��ʧȥ�ᾧˮ����ж�����ˮ�Ȼ��ܣ�
�۲���������������������ʽ����ʱ��Һ��pH������
| ������ | Fe��OH��3 | Fe��OH��2 | Co��OH��2 | Al��OH��3 |
| ��ʼ���� | 2.3 | 7.5 | 7.6 | 3.4 |
| ��ȫ���� | 4.1 | 9.7 | 9.2 | 5.2 |
��1���������¹����У��á����ᡱ����ԭ�����С�����������Ļ��ᡱֱ���ܽ⺬�ܷ��ϣ�����Ҫ�ŵ�Ϊ�����ж�������ŷţ���ֹ������Ⱦ���ֹ��Ʒ�л��������Σ�дһ�㣩��
��2������̼���Ƶ���pH��a��a�ķ�Χ��5.2-7.6��pH����a����ˣ������Լ�X����Һ��pH������2-3�������õ��Լ�XΪ���ᣮ
��3�����������3������ʵ�����������������Ũ������ȴ�ᾧ���ˣ�
��4���Ƶõ�CoCl2•6H2O���ѹ��ɵ�ԭ���ǽ��ͺ���¶ȣ���ֹ��Ʒ�ֽ⣮
��5��Ϊ�ⶨ��Ʒ��CoCl2•6H2O������ijͬѧ��һ��������Ʒ����ˮ���������м���������AgNO3��Һ�����ˣ�����������ɺ������������ͨ�����㷢�ֲ�Ʒ��CoCl2•6H2O��������������100%����ԭ���������Ʒ�к���NaCl���ʣ�CoCl2•6H2O���ʱʧȥ�˲��ֽᾧˮ��
��6����ʵ�����У�Ϊ�˴�������Ʒ�л�ô�����CoCl2•6H2O�������ƵõIJ�Ʒ�ܽ��������У�Ȼ����ˡ�������ʵ���������ô�����CoCl2•6H2O��
 ��
�� ��ù��������Ҫ�Ŀ����أ���ù���������ھ�����ˮ��õ���ù�ذ����ᣬ��ṹ��ʽ��ͼ��ʾ��
��ù��������Ҫ�Ŀ����أ���ù���������ھ�����ˮ��õ���ù�ذ����ᣬ��ṹ��ʽ��ͼ��ʾ��