��Ŀ����
(10��)�����£����ȡ0.1mol��L-1��HA��Һ��0.1mol��L-1��NaOH��Һ�������ϣ����Ի�Ϻ���Һ����ı仯������û��Һ��PH=8���Իش��������⣺
��1�������Һ��PH=8��ԭ�� �������ӷ���ʽ��ʾ����
��2�������Һ����ˮ�������c��H+�� 0.1mol��L-1NaOH��Һ����ˮ�������c��H+�����>���� <����=������
��3����������Һ��������ʽ�ľ�ȷ����������������֣�
c��Na+����c��A-��= mol��L-1��c��OH-����c��HA��= mol��L-1��
��4����֪NH4A��ҺΪ���ԣ���֪HA��Һ�ӵ�Na2CO3��Һ��������ų������ƶϣ�NH4��2CO3��Һ��PH ������ڡ���С�ڡ����ڡ���7����ͬ�¶��µ�Ũ�ȵ���������Һ��A��NH4HCO3 B��NH4A C����NH4��2SO4 D��NH4Cl����PH�ɴ�С��˳�������� ������ĸ����
��1��A-+H20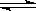 HA+OH- ��2��>��3��9.9��10-7
1.0��10-8��4������
ABDC
HA+OH- ��2��>��3��9.9��10-7
1.0��10-8��4������
ABDC
��������

 ѧ�����νӽ̲��Ͼ���ѧ������ϵ�д�
ѧ�����νӽ̲��Ͼ���ѧ������ϵ�д� Сѧ������ҵϵ�д�
Сѧ������ҵϵ�д�����10�֣��±���Ԫ�����ڱ���һ���֡��������е���ĸ�ֱ����һ�ֻ�ѧԪ�ء�
| A |
|
| |||||||||||||||
| B |
|
|
| C | D | E | F |
| |||||||||
| G | H | I | J |
| K | L |
| ||||||||||
|
|
|
|
|
| M |
| N |
|
|
|
|
|
|
|
|
|
|
�Իش��������⣺
��1����д��Ԫ��N�Ļ�̬ԭ�ӵ����Ų�ʽ ��
��2��D���⻯��������ˮ��ԭ��
��3��D��E�ĵ縺�ԵĴ�С�� �� ����Ԫ�ط��ű�ʾ��
��4����A��C��D�γɵ�ACD�����У����� ���Ҽ��� ���м���
��5��Ԫ��M�Ļ�����(MO2Cl2)���л��ϳ��п������������Ȼ��������������л��ﷴӦ����ش��������⣺
����Mͬ���ڵ�����Ԫ�صĻ�̬ԭ�����������������ԭ����ͬ��Ԫ���� __________����Ԫ�ط��ţ�������һ�ֽ����ľ����ṹ��ͼ��ʾ���þ����к��н���ԭ�ӵ���ĿΪ ��
��MO2Cl2������Ϊ���ɫҺ�壬����CCl4��CS2�Ȼ��ܣ��ݴ˿��ж�MO2Cl2��__________������ԡ��Ǽ��ԡ������ӡ�
���ڢٱ�����CH3OH����HCHO����CS2����CCl4�����л��ܼ��У�̼ԭ�Ӳ�ȡsp2�ӻ��ķ�����___________������ţ���CS2���ӵĿռ乹����__________��
����10�֣��±���Ԫ�����ڱ���һ���֡��������е���ĸ�ֱ����һ�ֻ�ѧԪ�ء�
| A | | | |||||||||||||||
| B | | | | C | D | E | F | | |||||||||
| G | H | I | J | | K | L | | ||||||||||
| | | | | | M | | N | | | | | | | | | | |
��1����д��Ԫ��N�Ļ�̬ԭ�ӵ����Ų�ʽ ��
��2��D���⻯��������ˮ��ԭ��
��3��D��E�ĵ縺�ԵĴ�С�� �� ����Ԫ�ط��ű�ʾ��
��4����A��C��D�γɵ�ACD�����У����� ���Ҽ��� ���м���
��5��Ԫ��M�Ļ�����(MO2Cl2)���л��ϳ��п������������Ȼ��������������л��ﷴӦ����ش��������⣺
����Mͬ���ڵ�����Ԫ�صĻ�̬ԭ�����������������ԭ����ͬ��Ԫ���� __________����Ԫ�ط��ţ�������һ�ֽ����ľ����ṹ��ͼ��ʾ���þ����к��н���ԭ�ӵ���ĿΪ ��
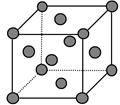
��MO2Cl2������Ϊ���ɫҺ�壬����CCl4��CS2�Ȼ��ܣ��ݴ˿��ж�MO2Cl2��__________������ԡ��Ǽ��ԡ������ӡ�
���ڢٱ�����CH3OH����HCHO����CS2����CCl4�����л��ܼ��У�̼ԭ�Ӳ�ȡsp2�ӻ��ķ�����___________������ţ���CS2���ӵĿռ乹����__________��
(14��) �����£���ijһԪ��HA��NaOH��Һ�������ϣ�������Һ��Ũ�Ⱥͻ�Ϻ�������Һ��pH���±���
| ʵ���� | HA���ʵ���Ũ�ȣ�mol/L�� | NaOH���ʵ���Ũ�ȣ�mol/L�� | ��Ϻ���Һ��pH |
| �� | 0.2 | 0.2 | pH = a |
| �� | C1 | 0.2 | pH = 7 |
| �� | 0.2 | 0.1 | pH >8 |
| �� | 0.1 | 0.1 | pH = 9 |
��1���������������ʵ���������Ӽ�����������������a��˵��HA��ǿ�ỹ�����ᡣ
__________________________________________
 _____________________________________��
_____________________________________����2���������������ʵ�����������������������C1�Ƿ�һ������0.2 mol/L________ ����Ϻ���Һ������Ũ��c(A-)��c(Na+)�Ĵ�С��ϵ��c(A-)_______ c(Na+) (��>��<��=)��
(3)�ӱ���ʵ����������HA��_______��(ǿ����)���û��Һ�е�����Ũ���ɴ�С��˳����______________________________________
��4��������Һ�У���ˮ�����c(H��) = mol��L-1��c (Na+)��c (A-)= mol��L-1��
��5����֪NH4A��ҺΪ���ԣ���֪��HA��Һ�ӵ�Na2CO3��Һ��������ų������ƶϣ�NH4��2CO3��Һ��pH 7 (��>��<��= )��
��6������ͬ�¶�����ͬŨ�ȵ���������Һ��
A��NH4HCO3 B��NH4A C��(NH4)2SO4 D��NH4Cl
��c(NH4+)�ɴ�С��˳������ ������ţ�
����10�֣��±���Ԫ�����ڱ���һ���֡��������е���ĸ�ֱ����һ�ֻ�ѧԪ�ء�
|
A |
|
|
|||||||||||||||
|
B |
|
|
|
C |
D |
E |
F |
|
|||||||||
|
G |
H |
I |
J |
|
K |
L |
|
||||||||||
|
|
|
|
|
|
M |
|
N |
|
|
|
|
|
|
|
|
|
|
�Իش��������⣺
��1����д��Ԫ��N�Ļ�̬ԭ�ӵ����Ų�ʽ ��
��2��D���⻯��������ˮ��ԭ��
��3��D��E�ĵ縺�ԵĴ�С�� �� ����Ԫ�ط��ű�ʾ��
��4����A��C��D�γɵ�ACD�����У����� ���Ҽ��� ���м���
��5��Ԫ��M�Ļ�����(MO2Cl2)���л��ϳ��п������������Ȼ��������������л��ﷴӦ����ش��������⣺
����Mͬ���ڵ�����Ԫ�صĻ�̬ԭ�����������������ԭ����ͬ��Ԫ���� __________����Ԫ�ط��ţ�������һ�ֽ����ľ����ṹ��ͼ��ʾ���þ����к��н���ԭ�ӵ���ĿΪ ��

��MO2Cl2������Ϊ���ɫҺ�壬����CCl4��CS2�Ȼ��ܣ��ݴ˿��ж�MO2Cl2��__________������ԡ��Ǽ��ԡ������ӡ�
���ڢٱ�����CH3OH����HCHO����CS2����CCl4�����л��ܼ��У�̼ԭ�Ӳ�ȡsp2�ӻ��ķ�����___________������ţ���CS2���ӵĿռ乹����__________��