��Ŀ����
 ij�����к��еĻ������ΪNi��Cu��Zn�Ļ���������ڶ�����̼������ȡ���ѣ��������Ҵ���ͬ���칹�壬���۵�-141.5�棬�е�-24.9�棬�ڼ��������¿ɷֽ�ɼ��顢���顢��ȩ�ȣ�
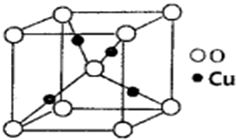
ij�����к��еĻ������ΪNi��Cu��Zn�Ļ���������ڶ�����̼������ȡ���ѣ��������Ҵ���ͬ���칹�壬���۵�-141.5�棬�е�-24.9�棬�ڼ��������¿ɷֽ�ɼ��顢���顢��ȩ�ȣ���1����ͼ��Cu��ij��������ľ����ṹʾ��ͼ��������λ��Ϊ
4
4
����2���Ҵ��ķе�ȼ��Ѹߣ�����Ҫԭ����
�Ҵ��γɷ��Ӽ����
�Ҵ��γɷ��Ӽ����
����3����ȩ������̼ԭ�ӹ�����ӻ�����Ϊ
sp2
sp2
����4��������ϻ�����A������ĵȵ����壬����Է�������Ϊ30.8����A���ɵڶ����������⻯���γɵĻ�̨�����A�Ỻ���ͷ�������ͬʱAת��Ϊ������B��B����ϩ�ĵȵ����壮������A�ĽṹʽΪ


5NA
5NA
����������1�����ݾ����ṹ�ж���ԭ����λ����
��2�����Ӱ�����ʵķе㣻
��3�����ݼ۲���ӶԻ�������ȷ�����ӻ���ʽ��
��4���γ���λ�����������пչ���µ��Ӷԣ�һ��BH2=NH2�����к���5���Ҽ���
��2�����Ӱ�����ʵķе㣻
��3�����ݼ۲���ӶԻ�������ȷ�����ӻ���ʽ��
��4���γ���λ�����������пչ���µ��Ӷԣ�һ��BH2=NH2�����к���5���Ҽ���
����⣺��1�����ݾ����ṹ֪����ԭ�ӵ���λ����4���ʴ�Ϊ��4��
��2���Ҵ����Ӽ����γ�����������ѷ��Ӽ䲻���γ����������Ĵ���ʱ���ʵķе����ߣ�
�ʴ�Ϊ���Ҵ��γɷ��Ӽ������
��3����ȩ��H2C=O��������̼ԭ���γ�3���Ҽ����¶Ե��ӣ��ӻ���ʽΪsp2�ӻ����ʴ�Ϊ��sp2��
��4��A���ɵڶ����������⻯���γɵĻ����A������ĵȵ����壬Ӧ����18�ĵ��ӣ�ΪBH3?NH3��B����ϩ�ĵȵ����壬ӦΪBH2=NH2��A��B���пչ����N���йµ��Ӷԣ����γ���λ��������A�ĽṹʽΪ ��һ��BH2=NH2�����к���5���Ҽ�������1mol������B�к��ЦҼ�����Ŀ5NA��
��һ��BH2=NH2�����к���5���Ҽ�������1mol������B�к��ЦҼ�����Ŀ5NA��
�ʴ�Ϊ�� ��5NA��
��5NA��
��2���Ҵ����Ӽ����γ�����������ѷ��Ӽ䲻���γ����������Ĵ���ʱ���ʵķе����ߣ�
�ʴ�Ϊ���Ҵ��γɷ��Ӽ������
��3����ȩ��H2C=O��������̼ԭ���γ�3���Ҽ����¶Ե��ӣ��ӻ���ʽΪsp2�ӻ����ʴ�Ϊ��sp2��
��4��A���ɵڶ����������⻯���γɵĻ����A������ĵȵ����壬Ӧ����18�ĵ��ӣ�ΪBH3?NH3��B����ϩ�ĵȵ����壬ӦΪBH2=NH2��A��B���пչ����N���йµ��Ӷԣ����γ���λ��������A�ĽṹʽΪ
 ��һ��BH2=NH2�����к���5���Ҽ�������1mol������B�к��ЦҼ�����Ŀ5NA��
��һ��BH2=NH2�����к���5���Ҽ�������1mol������B�к��ЦҼ�����Ŀ5NA���ʴ�Ϊ��
 ��5NA��
��5NA�����������⿼���Ϊ�ۺϣ��漰�ӻ���ʽ���ж��Լ���λ����֪ʶ����Ŀ�ѶȽϴ���Ҫע����λ�����γ������Լ��ӻ����͵��жϷ�����Ϊ�״��㣮

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ



