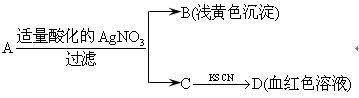
��Ŀ����
��6�֣���ͼ��һЩ�����ĵ��ʡ�������֮���ת����ϵͼ����Щ��Ӧ�еIJ������ʱ���ȥ����Ӧ�ٳ���Ӧ����Ұ�⺸�Ӹֹ죬AΪ�ճ������г����Ľ������ʣ� GΪ����ɫ���壬F�Ǻ�ˮ�к����ε���Ҫ�ɷ֣�JΪD��G��Ӧ�����γɵ�ˮ��Һ��
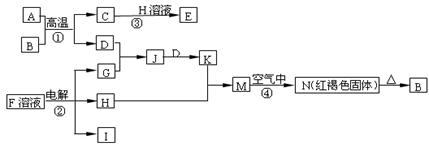
��ش��������⣺
��1��H�Ļ�ѧʽΪ_______________����2����Ӧ���ڹ�ҵ��ͨ����֮Ϊ__________��ҵ��
��3��д����Ӧ�۵����ӷ���ʽ ��
��4��д����Ӧ�ڵĻ�ѧ����ʽ ��

��ش��������⣺
��1��H�Ļ�ѧʽΪ_______________����2����Ӧ���ڹ�ҵ��ͨ����֮Ϊ__________��ҵ��
��3��д����Ӧ�۵����ӷ���ʽ ��
��4��д����Ӧ�ڵĻ�ѧ����ʽ ��
��6�֣�(1)NaOH (2)�ȼҵ
��3��Al2O3+2OH- =2AlO2-+H2O 2NaCl+2H2O=2NaOH+H2+Cl2
��3��Al2O3+2OH- =2AlO2-+H2O 2NaCl+2H2O=2NaOH+H2+Cl2
��

��ϰ��ϵ�д�
�����Ŀ
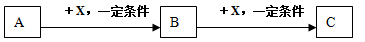
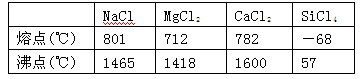
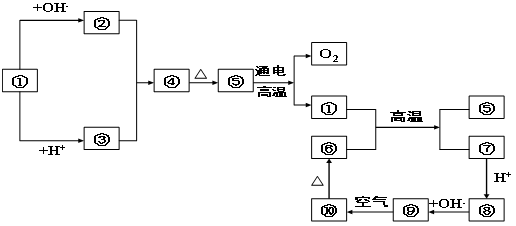

 ��
��
 ��ѧ��ѧ�еij������ʣ�����CΪO2��DΪC12��EΪFe���ʣ�����Ϊ��������Ǵ�������ת����ϵ����Ӧ�����ɵ�ˮ����Ҫ���������ȥ��
��ѧ��ѧ�еij������ʣ�����CΪO2��DΪC12��EΪFe���ʣ�����Ϊ��������Ǵ�������ת����ϵ����Ӧ�����ɵ�ˮ����Ҫ���������ȥ��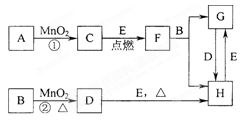
 �����ԡ����ԣ�ԭ�� ��
�����ԡ����ԣ�ԭ�� ��