��Ŀ����
��������(CeO2)��һ����Ҫ��ϡ�������ƽ�������ʾ�����������в��������ķϲ�����ĩ(��SiO2��Fe2O3��CeO2�Լ���������������ϡ�������)��ij�������Դ˷�ĩΪԭ�ϻ����棬���ʵ���������£�
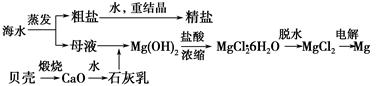
(1)ϴ������A��Ŀ����Ϊ��ȥ��________(�����ӷ���)������������Ƿ�ϴ���ķ�����___________________________________________________��
(2)�ڢڲ���Ӧ�����ӷ���ʽ��__________________________________������B����Ҫ�ɷ���________��
(3)��ȡ�Ƿ���ϡ��Ԫ�صij��÷�������֪������TBP��Ϊ��ȡ���ܽ������Ӵ�ˮ��Һ����ȡ������TBP________(��ܡ����ܡ�)��ˮ���ܡ�ʵ���ҽ�����ȡ����ʱ�õ�����Ҫ����������________���ձ�������������Ͳ�ȡ�
(4)ȡ���������еõ���Ce(OH)4��Ʒ0.536 g���������ܽ����0.100 0 mol��L��1 FeSO4����Һ�ζ��յ�(�汻��ԭΪCe3��)������25.00 mL����Һ���ò�Ʒ��Ce(OH)4����������Ϊ________��
��(1)Fe3����ȡ���һ��ϴ��Һ������KSCN��Һ���������ֺ�ɫ������ϴ������֮��δϴ��(���������𰸾���)
(2)2CeO2��H2O2��6H��=2Ce3����4H2O��O2����SiO2(���������𰸾���)
(3)���ܡ���Һ©��
(4)97.0%
����

 ��һ����ͬ���ɽ�����ϵ�д�
��һ����ͬ���ɽ�����ϵ�д� ������Ӧ���ϵ�д�
������Ӧ���ϵ�д� ��ʦ�㾦�ִʾ��ƪϵ�д�
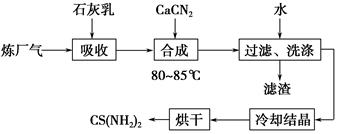
��ʦ�㾦�ִʾ��ƪϵ�д�����������(��Ҫ��Fe2O3��SiO2��Al2O3��MgO������)�Ʊ��������Ĺ����������£�
(1)�������������Ҫ�ʵ�������Ŀ���ǣ���������Ľ����ʣ���________��
(2)����ԭ���ǽ�Fe3��ת��ΪFe2����ͬʱFeS2������ΪSO42�����÷�Ӧ�����ӷ���ʽΪ________________________________________________________________________��
(3)Ϊ�ⶨ��������������Һ��Fe3�������Կ��Ƽ���FeS2������ʵ�鲽��Ϊ��ȷ��ȡһ���������������Һ����ƿ�У�����ϡ���ᡢ�Թ���SnCl2���ټ�HgCl2��ȥ������SnCl2���Զ�����������Ϊָʾ������K2Cr2O7����Һ�ζ����йط�Ӧ�Ļ�ѧ����ʽ���£�
2Fe3����Sn2����6Cl��=2Fe2����SnCl62��
Sn2����4Cl����2HgCl2=SnCl62����Hg2Cl2��
6Fe2����Cr2O72����14H��=6Fe3����2Cr3����7H2O
����SnCl2����������ⶨ��Fe3����________(�ƫ�ߡ�����ƫ�͡����䡱����ͬ)��
��������HgCl2����ⶨ��Fe3����________��
(4)�ٿ�ѡ��________(���Լ�)������Һ�к���Fe3��������Fe3����ԭ����________________________________________________________________________(�����ӷ���ʽ��ʾ)��
����֪����������������������ʽ����ʱ��Һ��pH���±���
| ������ | Fe(OH)3 | Al(OH)3 | Fe(OH)2 | Mg(OH)2 | Mn(OH)2 |
| ��ʼ���� | 2.7 | 3.8 | 7.5 | 9.4 | 8.3 |
| ��ȫ���� | 3.2 | 5.2 | 9.7 | 12.4 | 9.8 |
ʵ���ѡ�õ��Լ��У�ϡ���ᡢBa(NO3)2��Һ������KMnO4��Һ��NaOH��Һ��Ҫ���Ʊ������в������ж����塣
������ɡ����ˡ������Һģ���Ʊ���������ʵ�鲽�裺
a��������________________________________________________________��
b��������__________________________________________________________��
c�����룬ϴ�ӣ�
d����ɣ���ĥ��
���������Ҫ�ɷֿɱ�ʾΪFeO��Cr2O3��������MgO��Al2O3��Fe2O3�����ʣ��������Ը�����Ϊԭ���Ʊ��ظ���أ�K2Cr2O7��������ͼ��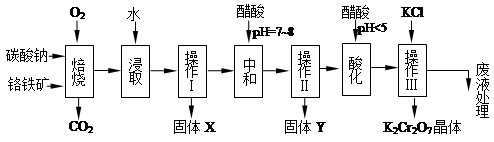
��֪����4FeO��Cr2O3+ 8Na2CO3+ 7O2 8Na2CrO4 + 2 Fe2O3 + 8CO2����
8Na2CrO4 + 2 Fe2O3 + 8CO2����
��Na2CO3 + Al2O3 2NaAlO2 + CO2������ Cr2O72��+ H2O
2NaAlO2 + CO2������ Cr2O72��+ H2O 2CrO42�� + 2H+
2CrO42�� + 2H+
��������ش��������⣺
��1������X����Ҫ����_________����д��ѧʽ����Ҫ����ữ��������Һ��pH�Ƿ����4.5��Ӧ��ʹ��__________����д�������Լ����ƣ���
��2���ữ�����ô��������ҺpH<5����Ŀ����_________________________________��
��3���������жಽ��ɣ����K2Cr2O7����IJ��������ǣ�����KCl���塢����Ũ���� �����ˡ�_______�����
��4���±���������ʵ��ܽ�����ݣ�����������Ӧ�Ļ�ѧ����ʽ�ǣ�Na2Cr2O7+2KCl ��K2Cr2O7��+2NaCl���÷�Ӧ����Һ���ܷ�����������_______________��
| ���� | �ܽ��/(g/100gˮ) | ||
| 0��C | 40��C | 80��C | |
| KCl | 28 | 40.1 | 51.3 |
| NaCl | 35.7 | 36.4 | 38 |
| K2Cr2O7 | 4.7 | 26.3 | 73 |
| Na2Cr2O7 | 163 | 215 | 376 |
��5������ƷY��Ҫ��������������������þ���������ܻ����P���������ʣ���ȷ����Y���������������dz�ȡn g��Ʒ���������______����д�Լ������ܽ⡢���ˡ���______����д�Լ������������ա���ȴ���������ø������m g ��������Ʒ��������������������Ϊ_______���ú�m��n�Ĵ���ʽ��ʾ����
��6�����۸��ж�����Cr3+���ȫ����ҵ������CrO3���������������ķ���֮һ�Ǹɷ��ⶾ����ú����ȫȼ�����ɵ�CO��ԭCrO3����ʵ������ģ����һ���̵�װ�����£�

CO�ɼ�����ˮ�Ƶã�ʵ�����ʱϨ��ƾ��Ƶ�˳����_________________________��