��Ŀ����
�����Ա����ж���ҽԺ�г������ᱵ���ֱ�����Ϊ�ڷ���Ӱ����ҽԺ���ȱ������ж���
��ʱ���������⣬����Ҫ���ж���θ�й�����������Һ��
��֪��Ksp(BaCO3)��5.1��10��9��Ksp(BaSO4)��1.1��10��10
�����ƶ���ȷ���ǣ� ��
��ʱ���������⣬����Ҫ���ж���θ�й�����������Һ��
��֪��Ksp(BaCO3)��5.1��10��9��Ksp(BaSO4)��1.1��10��10
�����ƶ���ȷ���ǣ� ��
| A������̼�ᱵ��Ϊ�ڷ���Ӱ��������ΪKsp(BaCO3)��Ksp(BaSO4) |
| B�����ȱ������ж�����ʱ����û�������ƣ�������̼������Һ���� |
| C��������c(Ba2��)��1.0��10��5 mol/L����Һʱ�������������ж� |
| D��������0.36 mol/L��Na2SO4��Һ���������ж�����ϴθ |
D
θҺ��Ϊ���ỷ��������̼������θҺ���ܽ⣬��A��B�������ᱵ���ڷ���Ӱ��˵�����ᱵ������ı�����Ũ���ǰ�ȫ�ģ���ʱc(Ba2��)��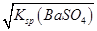 ��1.05��10��5 mol/L��1.0��10��5 mol/L��������c(Ba2��)��1.0��10��5 mol/L����Һʱ���������������ж���C������0.36 mol/L��Na2SO4��Һϴθʱ��θҺ��c(Ba2��)��Ksp(BaSO4)��c(SO42-)��(1.1��10��10��0.36) mol/L��3.1��10��10 mol/L��1.05��10��5 mol/L����D��ȷ��
��1.05��10��5 mol/L��1.0��10��5 mol/L��������c(Ba2��)��1.0��10��5 mol/L����Һʱ���������������ж���C������0.36 mol/L��Na2SO4��Һϴθʱ��θҺ��c(Ba2��)��Ksp(BaSO4)��c(SO42-)��(1.1��10��10��0.36) mol/L��3.1��10��10 mol/L��1.05��10��5 mol/L����D��ȷ��
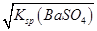 ��1.05��10��5 mol/L��1.0��10��5 mol/L��������c(Ba2��)��1.0��10��5 mol/L����Һʱ���������������ж���C������0.36 mol/L��Na2SO4��Һϴθʱ��θҺ��c(Ba2��)��Ksp(BaSO4)��c(SO42-)��(1.1��10��10��0.36) mol/L��3.1��10��10 mol/L��1.05��10��5 mol/L����D��ȷ��
��1.05��10��5 mol/L��1.0��10��5 mol/L��������c(Ba2��)��1.0��10��5 mol/L����Һʱ���������������ж���C������0.36 mol/L��Na2SO4��Һϴθʱ��θҺ��c(Ba2��)��Ksp(BaSO4)��c(SO42-)��(1.1��10��10��0.36) mol/L��3.1��10��10 mol/L��1.05��10��5 mol/L����D��ȷ��
��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
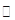 SO2 +2Cu��
SO2 +2Cu��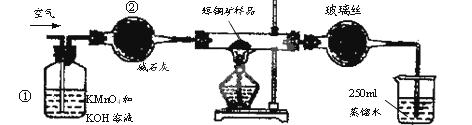
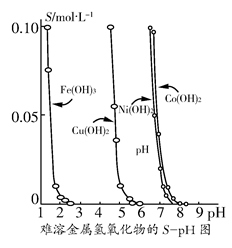
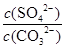 ��
��