��Ŀ����
(���8��)�ڲ��������У���¯��ԭ�Ϸ�������Ҫ��ӦΪ��
2Na2CO3+CaCO3+3SiO2 2Na2SiO3+CaSiO3+3CO2��
2Na2SiO3+CaSiO3+3CO2��
��1����ij��Ӧ��������״̬�²����磬������_____���塣
��2����������Ӧ������ڱ���������Ԫ�ص�ԭ�Ӱ뾶______��______(��Ԫ�ط���) ������ͬһ����Ԫ�ص�������__________________��
��3����������Ӧ���������ǿ�Ķ�����Ԫ�أ���ԭ�Ӻ�����______�ֲ�ͬ�ܼ��ĵ��ӣ��䵥����O2�г��ȼ�գ� ����ĵ���ʽΪ_____________��
��4����H2��O2������Ӧ�Ĺ�����ģ��ͼʾ���£���������ʾ��ѧ������
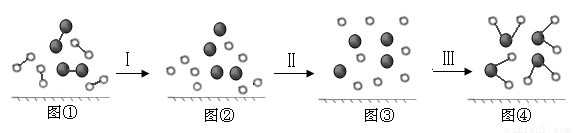
���й��̢���______���̣�����ȡ��������ȡ���û�������仯������ͼ���в������ɼ��Լ����ɵ�__________���ӣ�����ԡ��Ǽ��ԡ�����
��1����ԭ�� (1��)
��2����Si C (1��) ̼���� ����� (��2�֣���1��)
��3����4
 (��2�֣���1��)
(��2�֣���1��)
��4�������� (1��) ���� (1��)
����������1��.��������������״̬�²����磬��ԭ�Ӿ��塣
��2��.̼������ͬһ���������Ԫ�أ�ͬ�������϶���ԭ�Ӱ뾶���������Թ�ԭ�Ӱ뾶����̼ԭ�Ӱ뾶������ͬһ���ڵ���̼��Ϊ���Լ��ƺ衣
��3����������ǿ�����ƣ����ݹ���ԭ����֪����4�ֲ�ͬ�ܼ��ĵ��ӡ����������е�ȼ�ղ����ǹ������ƣ��������Ӽ��ͷǼ��Լ������ӻ����
��4��.����I�Ƕϼ����̣����ȡ�ˮ���ɼ��Լ����ɵļ��Է��ӡ�

 ����������������ϵ�д�
����������������ϵ�д� 2Na2SiO3+CaSiO3+3CO2��
2Na2SiO3+CaSiO3+3CO2��