��Ŀ����
 ��ҵ��������Ȼ������Ҫ�ɷ���CH4����H2O���и��������Ʊ��ϳ�����CO+H2����CO��H2��һ�����������Ʊ���ȩ�����ѵȶ����л����֪CH4��H2��CO��ȼ���ȣ���H���ֱ�Ϊ-890.3kJ?mol-1��-285.8kJ?mol-1��-283.0kJ?mol-1��18.0gˮ����Һ��ʱ�������仯Ϊ44.0kJ��
��ҵ��������Ȼ������Ҫ�ɷ���CH4����H2O���и��������Ʊ��ϳ�����CO+H2����CO��H2��һ�����������Ʊ���ȩ�����ѵȶ����л����֪CH4��H2��CO��ȼ���ȣ���H���ֱ�Ϊ-890.3kJ?mol-1��-285.8kJ?mol-1��-283.0kJ?mol-1��18.0gˮ����Һ��ʱ�������仯Ϊ44.0kJ����1��д��������ˮ�����ڸ����·�Ӧ�ƺϳ������Ȼ�ѧ����ʽ
CH4��g��+H2O��g��=CO��g��+3H2��g����H=+206.1kJ?mol-1
CH4��g��+H2O��g��=CO��g��+3H2��g����H=+206.1kJ?mol-1
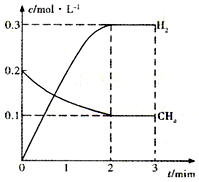
����2����һ�������£�������̶�Ϊ2L���ܱ������г���0.40mol CH4��g����0.60mol H2O��g�������CH4��g����H2��g�������ʵ���Ũ����ʱ��仯��������ͼ��ʾ��
3minʱ�ı���������Ӧ���е�4minʱ����ϵ�и����ʵ����ʵ������±���ʾ��
| t/min | n��CH4��/mol | n��H2O��/mol | n��CO��/mol | n��H2��/mol |
| 4 | 0.18 | 0.38 | 0.22 | 0.66 |
��
��
����������桱����Ӧ�����ƶ���3minʱ�ı�������������¶�
�����¶�
����3����֪�¶ȡ�ѹǿ��̼ˮ��[
| n(CH4) |
| n(H2O) |
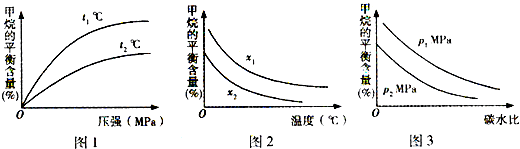
��ͼl�У�����������ʾ�¶ȵĹ�ϵ��t1
��
��
t2�����������������=������ͬ����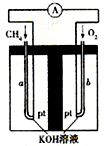
��ͼ2�У�����������ʾ̼ˮ�ȵĹ�ϵ��x1
��
��
x2����ͼ3�У�����������ʾѹǿ�Ĺ�ϵ��p1
��
��
p2����4������Ȼ�����������ʲ����뷴Ӧ��Ϊԭ�ϵ�ȼ�ϵ��ʾ��ͼ��ͼ��ʾ��
�ٷŵ�ʱ�������ĵ缫��ӦʽΪ
O2+4e-+2H2O=4OH-
O2+4e-+2H2O=4OH-
���ڼ���װ����ʢװ100.0mL 3.0mol?L-1 KOH��Һ���ŵ�ʱ���뷴Ӧ�������ڱ�״�������Ϊ8 960mL���ŵ���Ϻ������Һ�и�����Ũ�ȵĴ�С��ϵΪ
c��K+����c��HCO3-����c��CO32-����c��OH-����c��H+��
c��K+����c��HCO3-����c��CO32-����c��OH-����c��H+��
����������1������ȼ����д���Ȼ�ѧ����ʽ�����ø�˹���ɼ��㣻
��2������ͼ���֪3minʱ��ƽ��ʱ����ֵ�ƽ��Ũ�ȣ��ٸ���4minʱ�����Ũ�ȱ仯���жϸı��������
��3���ٸ��ݷ�Ӧ����ЧӦ�ж��¶ȶ�ƽ���ƶ���Ӱ�죬���ͼ��������
��̼ˮ��[
]ֵԽ��ƽ��ʱ�����ת����Խ�ͣ�����Խ�ߣ�
�۸���ѹǿ��ƽ���ƶ�Ӱ�죬���ͼ��������
��4��������������ԭ��Ӧ�������������ŵ��������������ӣ�
�ڼ������������ʵ����������������ɶ�����̼�����ʵ���������n��NaOH����n��CO2��������ϵ�жϷ�Ӧ�������������Һ�е�������ʵ������������ˮ���������жϣ�
��2������ͼ���֪3minʱ��ƽ��ʱ����ֵ�ƽ��Ũ�ȣ��ٸ���4minʱ�����Ũ�ȱ仯���жϸı��������
��3���ٸ��ݷ�Ӧ����ЧӦ�ж��¶ȶ�ƽ���ƶ���Ӱ�죬���ͼ��������
��̼ˮ��[
| n(CH4) |
| n(H2O) |
�۸���ѹǿ��ƽ���ƶ�Ӱ�죬���ͼ��������
��4��������������ԭ��Ӧ�������������ŵ��������������ӣ�
�ڼ������������ʵ����������������ɶ�����̼�����ʵ���������n��NaOH����n��CO2��������ϵ�жϷ�Ӧ�������������Һ�е�������ʵ������������ˮ���������жϣ�
����⣺��1����֪����H2��g��+
O2��g��=H2O��l����H=-285.8kJ?mol-1
��CO��g��+
O2��g��=CO2��g�� ����H=-283.0kJ?mol-1
��CH4��g��+2O2��g��=CO2��g��+2H2O��l������H=-890.3kJ?mol-1��
��H2O��g��=H2O��l����H=-44.0kJ?mol-1��
���ø�˹���ɽ���+��-��-3���ٿɵã�CH4��g��+H2O��g��=CO��g��+3H2��g��
��H=��-44.0kJ?mol-1��+��-890.3kJ?mol-1��-��-283.0kJ?mol-1��-3����-285.8kJ?mol-1��=+206.1 kJ?mol-1��
�ʴ�Ϊ��CH4��g��+H2O��g��=CO��g��+3H2��g����H=+206.1 kJ?mol-1��
��2������ͼ���֪3minʱ��ƽ��ʱ�����Ũ��Ϊ0.1mol/L��������Ũ��Ϊ0.3mol/L����
CH4��g��+H2O��g��=CO��g��+3H2��g��
��ʼ��mol/L����0.2 0.3 0 0
�仯��mol/L����0.1 0.1 0.1 0.3
ƽ�⣨mol/L����0.1 0.2 0.1 0.3
4minʱ�����Ũ��Ϊ0.09mol/L��Ũ�ȼ�С0.1-0.09=0.01
ˮ��Ũ��Ϊ0.19mol��Ũ�ȼ�С0.2-0.19=0.01��
CO��Ũ��Ϊ0.11��Ũ������0.11-0.1=0.01
������Ũ��Ϊ0.33��Ũ������0.33-0.3=0.03
Ũ�ȱ仯��֮��Ϊ1��1��1��3�����ڻ�ѧ������֮�ȣ�Ӧ�Ǹı��¶�ƽ��������Ӧ�����ƶ����÷�Ӧ����Ӧ�����ȷ�Ӧ����3min�ı�����Ϊ�����¶ȣ�
�ʴ�Ϊ�����������¶ȣ�
��3���ٸ÷�Ӧ����ӦΪ���ȷ�Ӧ�������¶�ƽ��������Ӧ�����ƶ�������ĺ������ͣ����¶�t1��t2���ʴ�Ϊ������
��̼ˮ��[
]ֵԽ��ƽ��ʱ�����ת����Խ�ͣ�����Խ�ߣ���x1��x2���ʴ�Ϊ������
�۸÷�Ӧ����Ӧ�������������ķ�Ӧ������ѹǿƽ�����淴Ӧ�����ƶ���ƽ��ʱ����ĺ������ͣ���p1��p2���ʴ�Ϊ������
��4��������������ԭ��Ӧ�������������ŵ��������������ӣ������缫��ӦʽΪ��O2+4e-+2H2O=4OH-���ʴ�Ϊ��O2+4e-+2H2O=4OH-��
�ڲ��뷴Ӧ�������ڱ�״�������Ϊ8960mL�����ʵ���Ϊ
=0.4mol�����ݵ���ת���غ��֪�����ɶ�����̼Ϊ
=0.2mol��n��NaOH��=0.1L��3.0mol?L-1=0.3mol��n��NaOH����n��CO2��=0.3mol��0.2mol=3��2������1��1��2��1֮�䣬������̼��ء�̼����أ���̼��ء�̼����ص����ʵ����ֱ�Ϊxmol��ymol����x+y=0.2��2x+y=0.3�����x=0.1��y=0.1����Һ��̼���ˮ�⣬̼�������ˮ����ڵ��룬��Һ�ʼ��ԣ���c��OH-����c��H+����̼�����ˮ��̶ȴ���̼���������c��HCO3-����c��CO32-����������Ũ�����ˮ��̶Ȳ���̼���Ũ��ԭ�������������ӣ���c��K+����c��HCO3-����c��CO32-����c��OH-����c��H+����
�ʴ�Ϊ��c��K+����c��HCO3-����c��CO32-����c��OH-����c��H+����
| 1 |
| 2 |
��CO��g��+
| 1 |
| 2 |
��CH4��g��+2O2��g��=CO2��g��+2H2O��l������H=-890.3kJ?mol-1��
��H2O��g��=H2O��l����H=-44.0kJ?mol-1��
���ø�˹���ɽ���+��-��-3���ٿɵã�CH4��g��+H2O��g��=CO��g��+3H2��g��
��H=��-44.0kJ?mol-1��+��-890.3kJ?mol-1��-��-283.0kJ?mol-1��-3����-285.8kJ?mol-1��=+206.1 kJ?mol-1��
�ʴ�Ϊ��CH4��g��+H2O��g��=CO��g��+3H2��g����H=+206.1 kJ?mol-1��
��2������ͼ���֪3minʱ��ƽ��ʱ�����Ũ��Ϊ0.1mol/L��������Ũ��Ϊ0.3mol/L����
CH4��g��+H2O��g��=CO��g��+3H2��g��
��ʼ��mol/L����0.2 0.3 0 0
�仯��mol/L����0.1 0.1 0.1 0.3
ƽ�⣨mol/L����0.1 0.2 0.1 0.3
4minʱ�����Ũ��Ϊ0.09mol/L��Ũ�ȼ�С0.1-0.09=0.01
ˮ��Ũ��Ϊ0.19mol��Ũ�ȼ�С0.2-0.19=0.01��
CO��Ũ��Ϊ0.11��Ũ������0.11-0.1=0.01
������Ũ��Ϊ0.33��Ũ������0.33-0.3=0.03
Ũ�ȱ仯��֮��Ϊ1��1��1��3�����ڻ�ѧ������֮�ȣ�Ӧ�Ǹı��¶�ƽ��������Ӧ�����ƶ����÷�Ӧ����Ӧ�����ȷ�Ӧ����3min�ı�����Ϊ�����¶ȣ�
�ʴ�Ϊ�����������¶ȣ�
��3���ٸ÷�Ӧ����ӦΪ���ȷ�Ӧ�������¶�ƽ��������Ӧ�����ƶ�������ĺ������ͣ����¶�t1��t2���ʴ�Ϊ������
��̼ˮ��[
| n(CH4) |
| n(H2O) |
�۸÷�Ӧ����Ӧ�������������ķ�Ӧ������ѹǿƽ�����淴Ӧ�����ƶ���ƽ��ʱ����ĺ������ͣ���p1��p2���ʴ�Ϊ������
��4��������������ԭ��Ӧ�������������ŵ��������������ӣ������缫��ӦʽΪ��O2+4e-+2H2O=4OH-���ʴ�Ϊ��O2+4e-+2H2O=4OH-��
�ڲ��뷴Ӧ�������ڱ�״�������Ϊ8960mL�����ʵ���Ϊ
| 8.96L |
| 22.4L/mol |
| 0.4mol��4 |
| 8 |
�ʴ�Ϊ��c��K+����c��HCO3-����c��CO32-����c��OH-����c��H+����
�����������ۺ��Խϴ��漰�Ȼ�ѧ����ʽ��д����ѧƽ��ͼ��ѧƽ���Ӱ�����ء���ѧƽ����㡢ԭ��ء���ѧ���㡢����Ũ�ȱȽϵȣ��Ѷ��еȣ��ǶԻ���֪ʶ��ѧ���������ۺϿ��飮

��ϰ��ϵ�д�
�����Ŀ
 2H2��O2����ѭ�����̷��������У�
2H2��O2����ѭ�����̷��������У� 2H2��O2����ѭ�����̷��������У�
2H2��O2����ѭ�����̷��������У�