��Ŀ����
(14��)��1����1 L AlCl3��FeCl3�����Һ�м��뺬a mol NaOH����Һʱ�������ij������ɴ����ֵ����������NaOH��Һ��������ʼ�ܽ⣬��ǰ������NaOH�����ﵽb molʱ���������ټ��٣���ԭ��Һ��Fe3+�����ʵ���Ũ��Ϊ ��AlCl3�����ʵ��� ��
��2����֪��2Fe3++2I����=��2Fe2++ I2������2Fe2++Br2 =��2Fe3++2Br��
������1mol FeI2��1.5mol FeBr2����Һ��ͨ��2mol Cl2����ʱ��������������������
�������ٵ���Һ��ͨ��3mol Cl2�������������Ӷ�Ӧ����������ֱ����������� ��
(3������m g��м�뺬�� n gHNO3��������Һǡ����ȫ��Ӧ���� m : n =" 1" : 2.7�� �÷�Ӧ�Ļ�ѧ����ʽΪ __________________________________________________�����軹ԭ����ֻ��һ�֣���ֻ����һ���Σ�
������ n g HNO3��ϡ������Һǡ��ʹ5.6g������ȫ�ܽ⣬���� n/4 gHNO3����ԭ��NO����������ԭ����� n �ķ�ΧΪ_________________________
��ij������п�����ᷴӦʱ�����ʵ���֮��Ϊ2:5����ʱ����Ļ�ԭ������____________
(14��)(1)��4a-3b ��/3��b-a (2) ��I- Fe2+ �� Fe3+ I2 Br2
��3���� 5Fe +12HNO3��5Fe(NO3)2 + N2�� + 6H2O ��16.8 �� n �� 25.2 ��N2O��NH4NO3
���������������1����AlCl3��FeCl3�����ʵ����ֱ���x��y��������йط�Ӧ�Ļ�ѧ����ʽ��AlCl3��3NaOH=Al(OH)3����3NaCl��FeCl3��3NaOH=Fe(OH)3����3NaCl��Al(OH)3��NaOH=NaAlO2��2H2O��֪��3x��3y��amol��x��bmol��amol�����y�� �����������ӵ�Ũ����
�����������ӵ�Ũ���� /L��
/L��
��2���ٸ���������ԭ��Ӧ�л�ԭ���Ļ�ԭ��ǿ�ڻ�ԭ����Ŀ�֪����ԭ��ǿ��˳����I����Fe2����Br����2mol�����õ�4mol���ӣ�1mol FeI2ʧȥ3mol������������������1mol�������ӡ�
��3mol�����õ�6mol���ӣ���Һ�е���������2mol���ӣ���������ʧȥ2.5mol�����Ի�������1.5mol�����ӣ�������������Fe3+��I2��Br2��
��3����������������ʵ���֮���� ��������������Ӧ����������������Ӧ�Ļ�ѧ����ʽ��5Fe +12HNO3��5Fe(NO3)2 + N2�� + 6H2O��
��������������Ӧ����������������Ӧ�Ļ�ѧ����ʽ��5Fe +12HNO3��5Fe(NO3)2 + N2�� + 6H2O��
�����������Ӧ�Ļ�ѧ����ʽ������Fe��4HNO3��Fe(NO3)3��NO����2H2O��3Fe��8HNO3��3Fe(NO3)2��2NO����4H2O�����ڷ�Ӧ����1/4�����ᱻ��ԭ�����Ը�������0.1mol��֪���������Сֵ�����ֵ�ֱ��� 16.8g��
16.8g��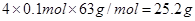 ����n��ȡֵ��Χ��16.8 �� n �� 25.2��
����n��ȡֵ��Χ��16.8 �� n �� 25.2��
����п�����ʵ�����2xmol����������4xmol����������п��2xmol������û�б���ԭ��������4xmol����ԭ��������5xmol��4xmol��xmol�����ݵ��ӵĵ�ʧ�غ��֪�������ڷ�Ӧ�еõ�4�����ӣ�����ԭ������N2O���������û�б���ԭ�����Ტû��ȫ����п���ӽ����������п����Ҳ������������泥���ʱ��Ӧ�Ļ�ѧ����ʽ����4Zn��10HNO3=4Zn(NO3)2��NH4NO3��3H2O�������ȷ�Ĵ���N2O��NH4NO3��
���㣺���������ӡ������Ӻ��������Ʒ�Ӧ���йؼ��㡢������ԭ��Ӧ���й��жϺͼ����Լ����������ᷴӦ���й��жϺͽ���
�����������ѶȽϴ��ؿ���ѧ�����������ۺ����ʣ���ѧ���ļ�����������˸��ߵ�Ҫ��������������ѧ���Ͻ�����˼ά��������ɢ˼ά�����Լ����Ӧ�����������������ѧ����ѧϰЧ�ʺ�Ӧ�������Լ��������⡢����������������ǿѧ����ѧϰ�����ġ�

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�(14��)��֪CO2(g)��H2(g) CO(g)��H2O(g)��ƽ�ⳣ�����¶ȱ仯���±���
CO(g)��H2O(g)��ƽ�ⳣ�����¶ȱ仯���±���
|
t/�� |
700 |
800 |
850 |
1 000 |
1 200 |
|
K |
2.6 |
1.7 |
1.0 |
0.9 |
0.6 |
��ش��������⣺
(1)��������Ӧ��________��Ӧ(ѡ����ȡ������ȡ�)��
(2)����ƽ����£���ƽ����________�ƶ���CO2ת����________������Ӧ����________(ѡ���������С�����䡱)��
(3)��850�淢��������Ӧ���Ա��е����ʵ���Ͷ����ݷ�Ӧ���У�����������Ӧ�����ƶ�����________(ѡ��A��B��C��D��E)
|
|
A |
B |
C |
D |
E |
|
n(CO2) |
3 |
1 |
0 |
1 |
1 |
|
n(H2) |
2 |
1 |
0 |
1 |
2 |
|
n(CO) |
1 |
2 |
3 |
0.5 |
3 |
|
n(H2O) |
5 |
2 |
3 |
2 |
1 |
(4)��850��ʱ�����淴Ӧ��CO2(g)��H2(g) CO(g)��H2O(g)���ڸ������ڸ����ʵ�Ũ�ȱ仯���£�
CO(g)��H2O(g)���ڸ������ڸ����ʵ�Ũ�ȱ仯���£�
|
ʱ��/min |
CO2 (mol/L) |
H2 (mol/L) |
CO (mol/L) |
H2O (mol/L) |
|
0 |
0.200 |
0.300 |
0 |
0 |
|
2 |
0.138 |
0.238 |
0.062 |
0.062 |
|
3 |
c1 |
c2 |
c3 |
c3 |
|
4 |
c1 |
c2 |
c3 |
c3 |
��3 min��4 minƽ���c3��______________��CO2��ת����Ϊ____________��
(14��)��֪CO2(g)��H2(g)CO(g)��H2O(g)��ƽ�ⳣ�����¶ȱ仯���±���
| t/�� | 700 | 800 | 850 | 1 000 | 1 200 |
| K | 2.6 | 1.7 | 1.0 | 0.9 | 0.6 |
��ش��������⣺
(1)��������Ӧ��________��Ӧ(ѡ����ȡ������ȡ�)��
(2)����ƽ����£���ƽ����________�ƶ���CO2ת����________������Ӧ����________(ѡ���������С�����䡱)��
(3)��850�淢��������Ӧ���Ա��е����ʵ���Ͷ����ݷ�Ӧ���У�����������Ӧ�����ƶ�����________(ѡ��A��B��C��D��E)
|
| A | B | C | D | E |
| n(CO2) | 3 | 1 | 0 | 1 | 1 |
| n(H2) | 2 | 1 | 0 | 1 | 2 |
| n(CO) | 1 | 2 | 3 | 0.5 | 3 |
| n(H2O) | 5 | 2 | 3 | 2 | 1 |
(4)��850��ʱ�����淴Ӧ��CO2(g)��H2(g)CO(g)��H2O(g)���ڸ������ڸ����ʵ�Ũ�ȱ仯���£�
| ʱ��/min | CO2 (mol/L) | H2 (mol/L) | CO (mol/L) | H2O (mol/L) |
| 0 | 0.200 | 0.300 | 0 | 0 |
| 2 | 0.138 | 0.238 | 0.062 | 0.062 |
| 3 | c1 | c2 | c3 | c3 |
| 4 | c1 | c2 | c3 | c3 |
��3 min��4 minƽ���c3��______________��CO2��ת����Ϊ____________��
 3H2(g)��N2(g)�����ﵽƽ�⡣��
3H2(g)��N2(g)�����ﵽƽ�⡣�� CH3OH(g)+H2O(g)����ͼ1��ʾ�÷�Ӧ����������(��λΪkJ��mol-1)�ı仯��
CH3OH(g)+H2O(g)����ͼ1��ʾ�÷�Ӧ����������(��λΪkJ��mol-1)�ı仯�� 