题目内容
下列各物质无论以何种比例混合,其氯元素的质量分数不变的是( )A.HClO4、KClO3
B.NaClO、KCl
C.KClO、KCl
D.NaCl、KCl
【答案】分析:根据物质中某元素的质量分数= ×100%,分别计算选项中氯元素的质量分数,氯元素的质量分数相同的符合题意.
×100%,分别计算选项中氯元素的质量分数,氯元素的质量分数相同的符合题意.
解答:解:A.HClO4中氯元素的质量分数为: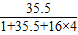 ×100%=35.32%;
×100%=35.32%;
KClO3中氯元素的质量分数为: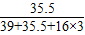 ×100%=28.98%;
×100%=28.98%;
B.NaClO中氯元素的质量分数为: ×100%=47.65%;
×100%=47.65%;
KCl中氯元素的质量分数为: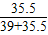 ×100%=47.65%;
×100%=47.65%;
C.KClO中氯元素的质量分数为: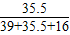 ×100%=39.23%;
×100%=39.23%;
KCl中氯元素的质量分数为: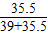 ×100%=47.65%;
×100%=47.65%;
D.NaCl中氯元素的质量分数为: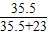 ×100%=60.68%;
×100%=60.68%;
KCl中氯元素的质量分数为: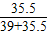 ×100%=47.65%;
×100%=47.65%;
由此可知,选项B符合题意;故选B.
点评:本题考查学生对物质中某元素的质量分数计算方法的掌握与应用的能力.
 ×100%,分别计算选项中氯元素的质量分数,氯元素的质量分数相同的符合题意.
×100%,分别计算选项中氯元素的质量分数,氯元素的质量分数相同的符合题意.解答:解:A.HClO4中氯元素的质量分数为:
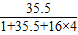 ×100%=35.32%;
×100%=35.32%;KClO3中氯元素的质量分数为:
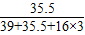 ×100%=28.98%;
×100%=28.98%;B.NaClO中氯元素的质量分数为:
 ×100%=47.65%;
×100%=47.65%;KCl中氯元素的质量分数为:
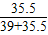 ×100%=47.65%;
×100%=47.65%; C.KClO中氯元素的质量分数为:
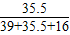 ×100%=39.23%;
×100%=39.23%;KCl中氯元素的质量分数为:
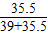 ×100%=47.65%;
×100%=47.65%; D.NaCl中氯元素的质量分数为:
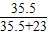 ×100%=60.68%;
×100%=60.68%;KCl中氯元素的质量分数为:
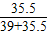 ×100%=47.65%;
×100%=47.65%;由此可知,选项B符合题意;故选B.
点评:本题考查学生对物质中某元素的质量分数计算方法的掌握与应用的能力.

练习册系列答案
相关题目