��Ŀ����
��Դ����ᷢչ����Ҫ��֤����ѧ����Դ��ʮ����Ҫ�Ĺ�ϵ������������й���Դ������Ĺ��ɣ���1�����ǰ�ֱ�Ӵ���Ȼȡ�õ���Դ��Ϊһ����Դ��һ����Դ�����ӹ�ȡ�õ���Դ��Ϊ������Դ��������Ŀǰ�������úͿ����IJ�����Դ ��ú�������ܣ�����Ȼ������̫���ܣ��ݷ��ܣ����в�����һ����Դ���ǣ�����ţ� ���������ڲ�����������Դ�� ��
��2���ҹ��������ú�����������úȼ���ŷŵ�SO2���������ꡱ���Խ������� ��Ӱ��ֲ��������Ӱ�죮Ϊ���ٴ�����Ⱦ��ij���糧�����ˡ�ʯ�ҹ���������ú�в�����ʯ��CaO����������SO2�����������Ϸ�Ӧ��д����������Ӧ�Ļ�ѧ����ʽ ��
��3��Ŀǰ�ҹ����������ʹ���Ҵ����ͣ���ʡ�ǻ��е���Ʒ�����أ���ÿ�궼�д����Ĺ�����ʳ--�»������֣��������ǿ����ó»����������ƾ��������Ϊ���»���
 ������
������ �ƾ�+������̼
�ƾ�+������̼��д������������ȡ�ƾ��Ļ�ѧ����ʽ ��
���𰸡���������1������Ҫ�ӹ���ֱ�Ӵ���Ȼ��ȡ�õ���Դ����Ϊһ����Դ��
��2������������������������ɵ��������������д����ѧ����ʽ��
��3��������Ϣ���»���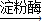 ������
������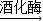 �ƾ�+������̼���������ǵĻ�ѧʽΪC6H12O6д����ѧ����ʽ��
�ƾ�+������̼���������ǵĻ�ѧʽΪC6H12O6д����ѧ����ʽ��
����⣺��1����ú������Ȼ������̫���ܣ��ݷ��ܣ��ȶ��Dz���Ҫ�ӹ���ֱ�Ӵ���Ȼ��ȡ�õ���Դ����Ϊһ����Դ����������Ҫ�����ӹ�ȡ�õ���Դ����Ϊ������Դ�����Т�ú������Ȼ�����ڲ���������Դ��һ����Դ�����ӹ�ȡ�õ���Դ����Ϊ������Դ�������ͣ����ܣ����ܣ��ƾ��ȶ��ǣ�
�ʴ�Ϊ���ڣ��٢ۣ�
��2������Խ������и�ʴ�ԡ�Ӱ��ֲ��������Ӱ�죮�������������������������ƣ��÷�Ӧ�Ļ�ѧ����ʽΪ��CaO+SO2=CaSO3��
�ʴ�Ϊ����ʴ�ԣ�CaO+SO2=CaSO3��
��3��������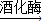 �ƾ�+������̼���û�ѧ����ʽ��ʾΪ��C6H12O6
�ƾ�+������̼���û�ѧ����ʽ��ʾΪ��C6H12O6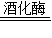 2C2H5OH+2CO2����
2C2H5OH+2CO2����
�ʴ�Ϊ��C6H12O6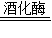 2C2H5OH+2CO2����
2C2H5OH+2CO2����
������������Ҫ���黯ѧ����ʽ����д���ѶȽϴ�
��2������������������������ɵ��������������д����ѧ����ʽ��
��3��������Ϣ���»���
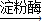 ������
������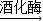 �ƾ�+������̼���������ǵĻ�ѧʽΪC6H12O6д����ѧ����ʽ��
�ƾ�+������̼���������ǵĻ�ѧʽΪC6H12O6д����ѧ����ʽ������⣺��1����ú������Ȼ������̫���ܣ��ݷ��ܣ��ȶ��Dz���Ҫ�ӹ���ֱ�Ӵ���Ȼ��ȡ�õ���Դ����Ϊһ����Դ����������Ҫ�����ӹ�ȡ�õ���Դ����Ϊ������Դ�����Т�ú������Ȼ�����ڲ���������Դ��һ����Դ�����ӹ�ȡ�õ���Դ����Ϊ������Դ�������ͣ����ܣ����ܣ��ƾ��ȶ��ǣ�
�ʴ�Ϊ���ڣ��٢ۣ�
��2������Խ������и�ʴ�ԡ�Ӱ��ֲ��������Ӱ�죮�������������������������ƣ��÷�Ӧ�Ļ�ѧ����ʽΪ��CaO+SO2=CaSO3��
�ʴ�Ϊ����ʴ�ԣ�CaO+SO2=CaSO3��
��3��������
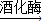 �ƾ�+������̼���û�ѧ����ʽ��ʾΪ��C6H12O6
�ƾ�+������̼���û�ѧ����ʽ��ʾΪ��C6H12O6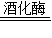 2C2H5OH+2CO2����
2C2H5OH+2CO2�����ʴ�Ϊ��C6H12O6
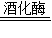 2C2H5OH+2CO2����
2C2H5OH+2CO2����������������Ҫ���黯ѧ����ʽ����д���ѶȽϴ�

��ϰ��ϵ�д�
 ��������ϵ�д�
��������ϵ�д�
�����Ŀ
 ������
������ �ƾ�+������̼
�ƾ�+������̼