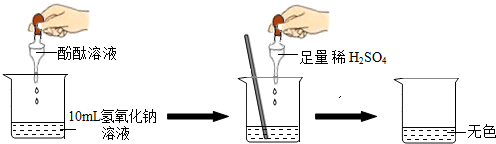
��Ŀ����
��2012?��̶����������ʵ��װ��ʾ��ͼ���ش��й����⣮
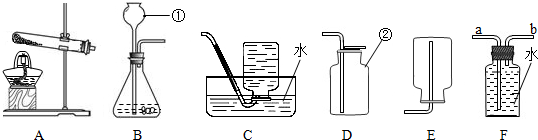
��1��Aװ����a������������
��2��ѡ��A��Bװ�ö����Ʊ���������
��3��װ��B��������ʵ������ȡ������̼�����г���©���¶˹ܿ������Һ���£���Һ�⣩��Ŀ���Ƿ�ֹ
����ٻ�ڣ��ڽ��룮�����ƶ�����̼�к���ˮ�������ɲ���

��1��Aװ����a������������
�ƾ���
�ƾ���
����2��ѡ��A��Bװ�ö����Ʊ���������
����
����
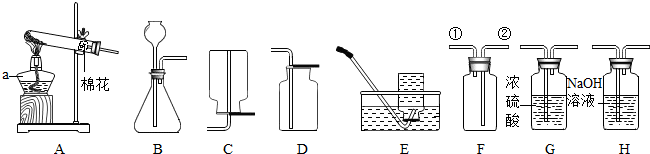
����Aװ����ȡ������Ļ�ѧ��Ӧ����ʽ��2KMnO4
2KMnO4+MnO2+O2��
| ||
2KMnO4
2KMnO4+MnO2+O2��
���ռ��������ѡ�õ�װ��Ϊ
| ||
E
E
������ţ����һ�ּ��ɣ�������ȡ������ʱ�Թ��е���û�з���ȼ�գ�����ȼ�յ���������������Ҫԭ�����¶�û�дﵽ�����Ż��
�¶�û�дﵽ�����Ż��
����3��װ��B��������ʵ������ȡ������̼�����г���©���¶˹ܿ������Һ���£���Һ�⣩��Ŀ���Ƿ�ֹ
���ɵ�����ӳ���©���ݳ�
���ɵ�����ӳ���©���ݳ�
������Fװ���ռ�������̼�������������
��
����ٻ�ڣ��ڽ��룮�����ƶ�����̼�к���ˮ�������ɲ���
G
G
������ţ�װ�ý��и����������1�����ݳ������������ƺ���;�жϣ�
��2������ABװ�õ��ص��������ȡ�����壻�ռ�װ�õ�ѡ��Ҫ��������ܶȺ��ܽ��ԣ��˽��ȼ��ȼ�յ�������
��3�����ݳ���©����������ȡ��ʹ��ʱ��ע��������������ݶ�����̼���ܶȱȿ��������������Ũ���������ˮ�Է�����
��2������ABװ�õ��ص��������ȡ�����壻�ռ�װ�õ�ѡ��Ҫ��������ܶȺ��ܽ��ԣ��˽��ȼ��ȼ�յ�������
��3�����ݳ���©����������ȡ��ʹ��ʱ��ע��������������ݶ�����̼���ܶȱȿ��������������Ũ���������ˮ�Է�����
����⣺��1�����ݳ������������ƺ���;�жϿ�֪a�ƾ��ƣ�
�ʴ�Ϊ���ƾ��ƣ�
��2�����������ü��ȸ��������ȡ�������ù���������Һ�Ͷ������̵Ļ���ﳣ������ȡ�����ȸ��������ȡ�����Ļ�ѧ����ʽ��2KMnO4
2KMnO4+MnO2+O2�����������ܶȱȿ�����������ˮ�����������ſ���������ˮ���ռ�����Aװ����ȡ����ʱ���Թ��е���û�з���ȼ�գ�����ȼ�յ���������������Ҫԭ���ǣ��¶�û�дﵽ�����Ż�㣻
�ʴ�Ϊ��������2KMnO4
2KMnO4+MnO2+O2����E���¶�û�дﵽ�����Ż�㣻
��3���������©���¹ܿڲ�����Һ������ʱ������������ͻ��©����������Գ���©���¶˹ܿ�Ҫ����Һ�����£��������̼���ܶȱȿ����������Fװ���ռ�������̼����������Ӣڵ��ܽ��룬�����Ӣٵ����ų���Ũ���������ˮ�ԣ���������ƶ�����̼�к���ˮ�������ɲ���Gװ�ý��и��
�ʴ�Ϊ�����ɵ�����ӳ���©���ݳ����ڣ�G��
�ʴ�Ϊ���ƾ��ƣ�
��2�����������ü��ȸ��������ȡ�������ù���������Һ�Ͷ������̵Ļ���ﳣ������ȡ�����ȸ��������ȡ�����Ļ�ѧ����ʽ��2KMnO4
| ||
�ʴ�Ϊ��������2KMnO4
| ||
��3���������©���¹ܿڲ�����Һ������ʱ������������ͻ��©����������Գ���©���¶˹ܿ�Ҫ����Һ�����£��������̼���ܶȱȿ����������Fװ���ռ�������̼����������Ӣڵ��ܽ��룬�����Ӣٵ����ų���Ũ���������ˮ�ԣ���������ƶ�����̼�к���ˮ�������ɲ���Gװ�ý��и��
�ʴ�Ϊ�����ɵ�����ӳ���©���ݳ����ڣ�G��
�������������ڿ���ͬѧ�Ƕ���������ȡ��Ӧԭ����������������������ȷ���ռ�����������������ܸ���ȼ�յ��������ʵ�����⣬�����һ������ѧ������������ȡ���ռ������Ĺ��̡���ϰ��

��ϰ��ϵ�д�
 �������ͬ������ϵ�д�
�������ͬ������ϵ�д�
�����Ŀ
 ��2012?̩����ģ�⣩�±���������ڲ�ͬ�¶�ʱ���ܽ�ȣ�
��2012?̩����ģ�⣩�±���������ڲ�ͬ�¶�ʱ���ܽ�ȣ�