��Ŀ����
����Ŀ��ijУ��ѧ��ȤС����д��Σ�������ɳ���ᴿʵ�飬���������þ�������100g20%���Ȼ�����Һ��
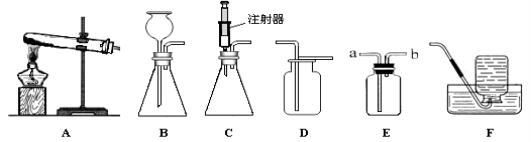
ʵ��һ����ͼ1�Ǽ�ͬѧ���д����ᴿʵ��IJ���ʾ��ͼ��
��1���������е�һ�����Դ����� ��
��2�������ݶ�Ӧ��ʵ���У�ֹͣ���ȵ�ǡ��ʱ���� ��
��3����ͬѧʵ����ȱ�ٹ��˲������ڴ˲����в������������� ��
ʵ�������ͬѧ���ᴿ�õ��ľ�������100g20%���Ȼ�����Һ��
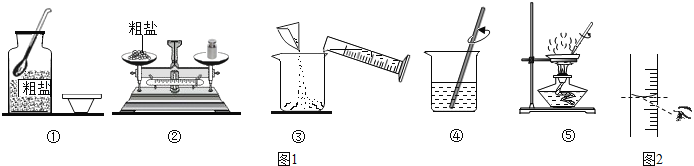
��4������ʱ������ͼ2�ṩ�������⣬����Ҫ�Լ�ƿ�����Ӻ� ����һ���������ƣ���
��5����������������ȷ��������ͼ2��ʾ�۲췽ʽ��ȡˮ����������Һ�������������� �����������������=����20%��
���𰸡���1��ƿ��û�е��ţ�
��2�����������г��ֽ϶����Ĺ���ʱ��
��3����������4����ͷ�ιܣ���5������
��������
���������������ѧ֪ʶ��������Ϣ֪��ʵ��һ����ͼ1�Ǽ�ͬѧ���д����ᴿʵ��IJ���ʾ��ͼ����1���������е�һ�����Դ�����ƿ��û�е��ţ���2����������Ӧ��ʵ���У�ֹͣ���ȵ�ǡ��ʱ���Ǵ��������г��ֽ϶����Ĺ���ʱ����3����ͬѧʵ����ȱ�ٹ��˲������ڴ˲����в�������������������ʵ�������ͬѧ���ᴿ�õ��ľ�������100g20%���Ȼ�����Һ����4������ʱ������ͼ2�ṩ�������⣬����Ҫ�Լ�ƿ�����Ӻͽ�ͷ�ιܣ���5����������������ȷ��������ͼ2��ʾ�۲췽ʽ��ȡˮ����������Һ����������������20%��
�������������Σ�������ɳ���ᴿʵ����

 �Ƹ������������ϵ�д�
�Ƹ������������ϵ�д�����Ŀ���±���Ca��OH��2��NaOH���ܽ�����ݣ���ش��������⣺
�¶�/�� | 0 | 20 | 40 | 60 | 80 | 100 | |
�ܽ��/g | Ca��OH��2 | 0.19 | 0.17 | 0.14 | 0.12 | 0.09 | 0.08 |
NaOH | 31 | 91 | 111 | 129 | 313 | 336 | |
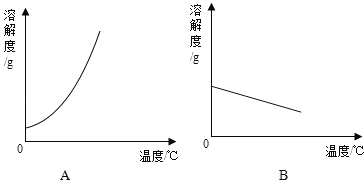
��1�������ϱ����ݣ�����Ca��OH��2��NaOH���ܽ�����ߣ���ͼ��ʾ�ܱ�ʾNaOH�ܽ�����ߵ��� _________ ����A��B����
��2��Ҫ���һƿ�ӽ����͵�Ca��OH��2��Һ��ɱ�����Һ�������ʩ�У��������������ƣ��������¶ȣ��������¶ȣ�������ˮ��������ˮ���ٻָ���ԭ�¶ȣ���������ʯ�ң�
���д�ʩ��ȷ���� _________��
A���ڢܢ� B���ۢ� C���٢ۢݢ� D���٢ڢݢ�
��3��20��ʱ��191g����NaOH��Һ������10gˮ���ٽ��µ�20����������NaOH���������Ϊ _________ ��
��4������20��ʱCa��OH��2�ı�����Һ������Һ���������м���һ����CaO��õ�����Һ������Һ������ʱ��Һ�����ʵ��������� �� _________ �ף����������������=������
��5������60��ʱ��Ca��OH��2��NaOH�������ʵı�����Һ����Ҫ�õ��ϴ�����NaOH���壬Ӧ��ȡ������������ _________ ��
��6��20��ʱ�����ⶨNaOH��Һ��pH�����Ƚ�pH��ֽ������ˮ��ʪ���ٽ��вⶨ����������Һ��pH _________ ���ƫ����ƫС������Ӱ�족����