��Ŀ����
�ݱ������ҹ��������ʴ��� ����ʧռ����������ֵ��GNP����4%��
��1����ͼ��ʾ����ʢˮ���Թ��з�һ���ྻ���������ô�U�ܵĽ���������U����ˮ�洦��ͬһ�߶ȣ������۲쵽U���ڵ�a��Һ�� ��������������½������䡱���������������ԭ���� 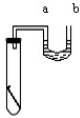 ��
��
��2��������ݲ�ͬԭ���������ֹ������ʴ������������ʩ��
�� ��
��3����д����ϡ�����ȥ����Ļ�ѧ��Ӧ����ʽ�� ��
��1����ͼ��ʾ����ʢˮ���Թ��з�һ���ྻ���������ô�U�ܵĽ���������U����ˮ�洦��ͬһ�߶ȣ������۲쵽U���ڵ�a��Һ��

��2��������ݲ�ͬԭ���������ֹ������ʴ������������ʩ��
��
��3����д����ϡ�����ȥ����Ļ�ѧ��Ӧ����ʽ��
���㣺������ʴ�������������,��Ļ�ѧ����,��д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ
ר�⣺�������������
�������������е�֪ʶ���з�����������ˮ�������Ӵ�ʱ�����⣬��������ƻ���������������ݴ˽��
����⣺��1�����������������������Թ��ڵ�ѹǿ��С���ڴ���ѹ�������ߣ�a��Һ����������������������������������Թ�����ѹ��С����������ѹ�����£�a��Һ��������
��2����������ƻ�����������������������ı���ˢ�ᡢͿ�ͣ����Ǹı����ڲ��Ľṹ�����ˢ�ᣬͿ�ͣ�
��3���������������ᷴӦ������������ˮ�����Fe2O3+3H2SO4=Fe2��SO4��3+3H2O��
��2����������ƻ�����������������������ı���ˢ�ᡢͿ�ͣ����Ǹı����ڲ��Ľṹ�����ˢ�ᣬͿ�ͣ�
��3���������������ᷴӦ������������ˮ�����Fe2O3+3H2SO4=Fe2��SO4��3+3H2O��
���������⿼����������������Լ�����Ĵ�ʩ������������������е�֪ʶ���У�

��ϰ��ϵ�д�
�����Ŀ
���������У���������������ǣ�������
| A��������أ�KMnO4�� |
| B�����������ף�P2O5�� |
| C��ˮ��H2O�� |
| D������þ��MgO�� |