��Ŀ����
�ṩ�������ϣ�
��1��
��2��CuO+H2SO4==CuSO4+H2O
��3��Cu��OH��2+H2SO4==CuSO4+2H2O
������3����Ļ�ѧ��ȤС������÷�ͭм��ȡ����ͭ����λͬѧ����Ʒ������£�
����һ��Cu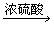 CuSO4
CuSO4
��������Cu
��������Cu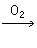 CuO
CuO CuSO4
CuSO4
�����ģ�Cu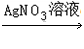 Cu��NO3��2
Cu��NO3��2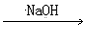 Cu��OH��2
Cu��OH��2 CuSO4
CuSO4
������ۡ����á������������ȷ���ȷ��һ�����ŵķ��������������ۣ�
��1�����ŷ�����_________��
��2��������ۣ������Ͽ��У����������в��������Ⱦ��ֻ��ϡ����һ���Լ����ɱ��ͣ��Ҳ�����
��1��

��2��CuO+H2SO4==CuSO4+H2O
��3��Cu��OH��2+H2SO4==CuSO4+2H2O
������3����Ļ�ѧ��ȤС������÷�ͭм��ȡ����ͭ����λͬѧ����Ʒ������£�
����һ��Cu
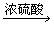 CuSO4
CuSO4��������Cu

��������Cu
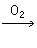 CuO
CuO CuSO4
CuSO4�����ģ�Cu
 Cu��NO3��2
Cu��NO3��2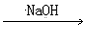 Cu��OH��2
Cu��OH��2 CuSO4
CuSO4������ۡ����á������������ȷ���ȷ��һ�����ŵķ��������������ۣ�
��1�����ŷ�����_________��
��2��������ۣ������Ͽ��У����������в��������Ⱦ��ֻ��ϡ����һ���Լ����ɱ��ͣ��Ҳ�����
������

��ϰ��ϵ�д�
 Ӣ�żƻ���ĩ����ϵ�д�
Ӣ�żƻ���ĩ����ϵ�д�
�����Ŀ
 CuSO4+SO2��+2H2O
CuSO4+SO2��+2H2O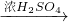 CuSO4
CuSO4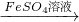 ��������Cu
��������Cu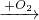 CuO
CuO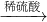 CuSO4
CuSO4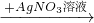 Cu��NO3��2
Cu��NO3��2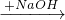 Cu��OH��2
Cu��OH��2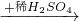 CuSO4
CuSO4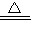 CuSO4+SO2��+2H2O
CuSO4+SO2��+2H2O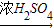 CuSO4
CuSO4 ��������Cu
��������Cu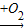 CuO
CuO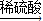 CuSO4
CuSO4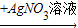 Cu��NO3��2
Cu��NO3��2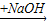 Cu��OH��2
Cu��OH��2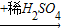 CuSO4
CuSO4