��Ŀ����
ʵ����ѧϰ��ѧ��һ����Ҫ;�����������ͼ�����ش��й����⣺

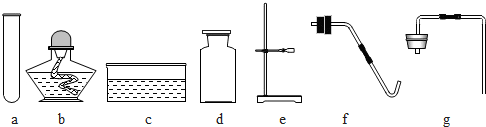
(1)д��ָ�����������ƣ���_______����________��
(2)����KMnO4��ȡ���ռ�O2��Ӧѡ�õ��������٢ܢޢ����________(�����)����Ӧ�Ļ�ѧ����ʽΪ_________________��
(3)ʵ������ȡCO2�Ļ�ѧ����ʽΪ_______________����ѡ��_______������CO2��O2��
(4)ʵ������ȡCO2��ʵ������ȡH2��Ƚϣ�����˵����ȷ����______(�����)��
�����巢��װ����ͬ �ڶ����������ſ������ռ� �۷�Ӧ������ͬ �ܻ�ѧ��Ӧ��������ͬ
(2)����KMnO4��ȡ���ռ�O2��Ӧѡ�õ��������٢ܢޢ����________(�����)����Ӧ�Ļ�ѧ����ʽΪ_________________��
(3)ʵ������ȡCO2�Ļ�ѧ����ʽΪ_______________����ѡ��_______������CO2��O2��
(4)ʵ������ȡCO2��ʵ������ȡH2��Ƚϣ�����˵����ȷ����______(�����)��
�����巢��װ����ͬ �ڶ����������ſ������ռ� �۷�Ӧ������ͬ �ܻ�ѧ��Ӧ��������ͬ
(1)����̨������ƿ
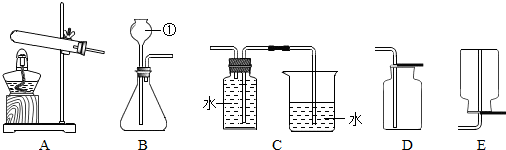
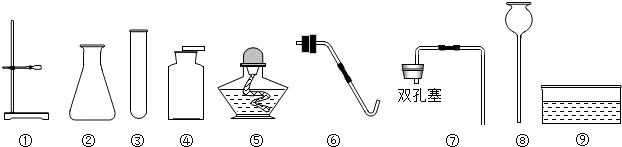
(2)�ۢݣ�2KMnO4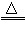 K2MnO4+MnO2+O2��
K2MnO4+MnO2+O2��
(3)CaCO3+2HCl==CaCl2+CO2��+H2O�������ʯ��ˮ(��ʯ����Һ��ȼ�ŵ�ľ����)
(4)�٢�
(2)�ۢݣ�2KMnO4
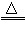 K2MnO4+MnO2+O2��
K2MnO4+MnO2+O2��(3)CaCO3+2HCl==CaCl2+CO2��+H2O�������ʯ��ˮ(��ʯ����Һ��ȼ�ŵ�ľ����)
(4)�٢�

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ

 ʵ����ѧϰ��ѧ��һ����Ҫ;�������ּӳ�װ�ã���
ʵ����ѧϰ��ѧ��һ����Ҫ;�������ּӳ�װ�ã���