��Ŀ����
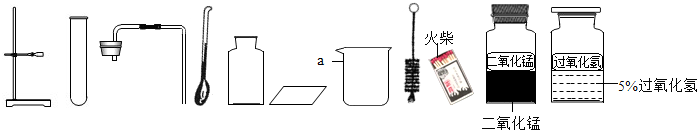
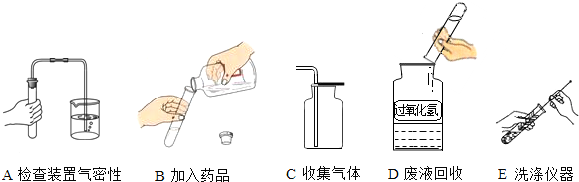
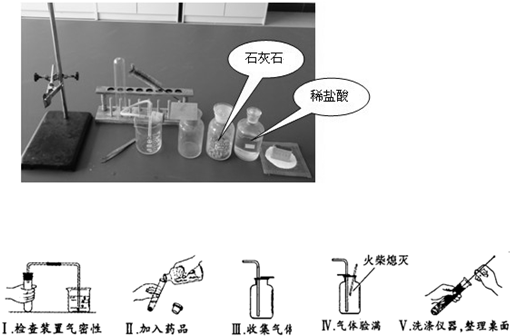
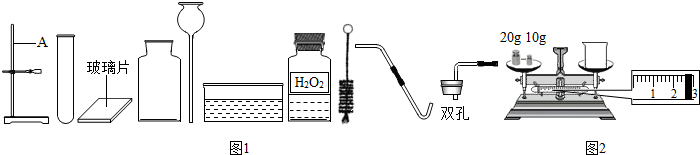
ijУ�����п���ѧʵ����������������ĸ����⣺�ٴ����ᴿ ����Ļ�ѧ���� �۶�����̼����ȡ���ռ������� ����������ȡ���ռ�������������ķ������ɿ�����ǩȷ�����⣬С��ͬѧ��ǩ���ʦ������������ͼ������ҩƷ��ʵ��̨ǰ����ش���1��������С����ɸ�ʵ����Ҫ�������̵�ʾ��ͼ�������ֱ���ÿ�������ȷ��1�֣�����5�֣�ʵ����Ϻ�С������3�֣����ҳ�������ʧ�ֵIJ�����˵������ԭ______

��2����������������ҩƷ��ѡ����Ҳ�������һ�ֳ��������ʵ������ȡ����ѧ����ʽΪ��______ 2H2O+O2��
���𰸡���������1�����ʵ������������Կ���С���IJ����г�������������ȡҺʱƿ��δ���š�����ʱ��ȼ�ŵĻ�����˼���ƿ�ڣ�
��2��������̼�ķ���װ��Ϊ��Һ��ϲ����ȵ�װ�ã����Ը��ݹ�������ΪҺ�塢��������Ϊ�����ҷ�Ӧ������ȣ��õ��������ù�������Ͷ���������ȡ�������ø��������������Ҫ�õ����ȣ����Ի��貹�������Ϊ�ƾ��ƣ�
����⣺��1��С���IJ����г�������������ȡҺʱƿ��δ���š�����ʱ��ȼ�ŵĻ�����˼���ƿ�ڣ��ʴ�Ϊ��ȡҺ��ʱƿ��δ���ţ� ����ʱ���ܰ�ȼ�ŵĻ�����뼯��ƿ�ڣ�
��2��ʵ�����ù�������������Ϊ��Һ�����͵ķ���װ�ã���Ӧԭ��Ϊ��2H2O2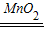 2H2O+O2�����ø��������������Ҫ�ø�������ȵ�װ�ã����Ի��貹��ƾ��ƣ�
2H2O+O2�����ø��������������Ҫ�ø�������ȵ�װ�ã����Ի��貹��ƾ��ƣ�
�ʴ�Ϊ����1��ȡҺ��ʱƿ��δ���ţ� ����ʱ���ܰ�ȼ�ŵĻ�����뼯��ƿ��
��2��2H2O2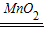 2H2O+O2�� �ƾ���
2H2O+O2�� �ƾ���
���������л�ѧ���������ʵ�����Ʒ���Ҫ��ҩƷ����Ӧԭ��������װ�á��ռ�װ�á����顢��������ʵ�������ע������ȷ�����бȽϡ����ɣ������ܽ�ʵ������ȡ�����һ��˼·�ͷ�����
��2��������̼�ķ���װ��Ϊ��Һ��ϲ����ȵ�װ�ã����Ը��ݹ�������ΪҺ�塢��������Ϊ�����ҷ�Ӧ������ȣ��õ��������ù�������Ͷ���������ȡ�������ø��������������Ҫ�õ����ȣ����Ի��貹�������Ϊ�ƾ��ƣ�
����⣺��1��С���IJ����г�������������ȡҺʱƿ��δ���š�����ʱ��ȼ�ŵĻ�����˼���ƿ�ڣ��ʴ�Ϊ��ȡҺ��ʱƿ��δ���ţ� ����ʱ���ܰ�ȼ�ŵĻ�����뼯��ƿ�ڣ�
��2��ʵ�����ù�������������Ϊ��Һ�����͵ķ���װ�ã���Ӧԭ��Ϊ��2H2O2
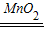 2H2O+O2�����ø��������������Ҫ�ø�������ȵ�װ�ã����Ի��貹��ƾ��ƣ�
2H2O+O2�����ø��������������Ҫ�ø�������ȵ�װ�ã����Ի��貹��ƾ��ƣ��ʴ�Ϊ����1��ȡҺ��ʱƿ��δ���ţ� ����ʱ���ܰ�ȼ�ŵĻ�����뼯��ƿ��
��2��2H2O2
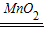 2H2O+O2�� �ƾ���
2H2O+O2�� �ƾ������������л�ѧ���������ʵ�����Ʒ���Ҫ��ҩƷ����Ӧԭ��������װ�á��ռ�װ�á����顢��������ʵ�������ע������ȷ�����бȽϡ����ɣ������ܽ�ʵ������ȡ�����һ��˼·�ͷ�����

��ϰ��ϵ�д�
�����Ŀ