题目内容
王腾同学以不同的化学思维方式或者规律给下列化学反应的类型进行分类,你认为分类合理的一组是:( )
①CaO+H2O═Ca(OH)2;
②Mg(OH)2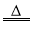 MgO+H2O;
MgO+H2O;
③Ba(OH)2+2HCl═BaCl2+2H2O;
④Zn+H2SO4═ZnSO4+H2↑;
⑤2Mg+O2 2MgO。
2MgO。
A.属于化合反应的是①② B.属于复分解反应的是③④
C.属于有热量放出的反应的是②③ D.属于有元素化合价变化的是④⑤
练习册系列答案
 字词句段篇系列答案
字词句段篇系列答案
相关题目
下列化学方程式及反应类型均正确的是
化学方程式 | 反应类型 | |
A | CO+ O2 | 化合反应 |
B | 2KMnO4 | 分解反应 |
C | 2Ag + H2SO4 | 置换反应 |
D | CuSO4+ BaCl2 | 复分解反应 |
A. A B. B C. C D. D


 CO2
CO2 K2MnO2+MnO2+2O2↑
K2MnO2+MnO2+2O2↑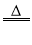 Cu + H2O B.2Mg + CO2
Cu + H2O B.2Mg + CO2 2MgO + C
2MgO + C