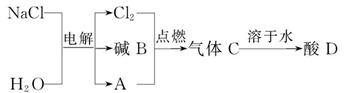
��Ŀ����
�κ��к�Cl-��HCO3-��Na+��K+��Mg2+��Ca2+�ȣ�ˮ��������ͷ��ξ���(��NaCl������)��
��1��д����Mg2+��HCO3-�γɻ�����Ļ�ѧʽ ,Ca2+������_________��
��2�������������Ϸ�������ˮ���Ȼ��Ƶ� ��Һ(����͡������͡�)�����ܼ��� ��
��3��ȡ��������ĺ�ˮ������ ���ж���ˮ��Ӳˮ��˵�����н϶��Mg2����Ca2����
��4������Ȼ���л�õĴ����к��н϶�����ʣ���ȥ��������ɳ�Ȳ��������ʵIJ�������Ϊ �� ��������ϴ�ӣ������������У����������ڳ��ֽ϶����ʱ��Ӧ��________(����ĸ)��
A��������������ˮ�֣�����ȥ�ƾ��� B����ȥ�ƾ��ƣ��ò�������������
��1��Mg(HCO3)2 ������ ��2�� ���ͣ� H2O
��3������ˮ�� ��4�� �ܽ� ���� B
���������������1������Ԫ�أ���ԭ���ţ��Ļ��ϼ�д��ѧʽ���ھ�����ǰ�����������ϼ۵�д��ǰ�棬�����ϼ۵�д�ں��棩�����棨���������ϼۻ�Ϊ�����ȣ��ٽ���д�ڶ�ӦԪ�ط��ŵ����½ţ�����Mg2+��HCO3-�γɻ�����Ļ�ѧʽ��Mg(HCO3)2����Ԫ�ط������Ͻ��������ֺ������������ӷ��ŵı�ʾ��������Ca2+�����ƣ�������
��2����Һ��������ͷ��ξ��壬�����DZ�����Һ���ܼ���ˮ
��3������Ӳˮ����ˮ�ķ������������ˮ
��4�����ε��ᴿʵ�鲽�裺�ܽ⡢���ˡ����������������ڳ��ֽ϶����ʱ��Ӧ��ֹͣ���ȣ��������Ȱ������ɣ�ͬʱΪ�˷�ֹ����ɽ����ò������������裬����ѡB
���㣺��ѧʽ����д����Һ��Ӳˮ����ˮ�ļ��𣬴��ε��ᴿ

 ��У����ϵ�д�
��У����ϵ�д�С�����˸������ͣ�����һ�����ʡ���Ƥ������ӡ����ͼ��ʾ�ı�ǩ�����ᵽһ�ɳ�ζ��
| ̼�����NH4HCO3 ���أ�50 kg ����������17.38% ������ѧ��ҵƷ��˾ |
����˸�����˵��
(1)���������______________�ʣ��ܴ�ʹ���ᆬҶ����ïʢ��ҶɫŨ�̣�ÿ�������к���Ԫ������Ϊ________________kg��
(2)�û���____________ʱ���ֽ⣬��ʩ��ʱ������__________�����ʻ��á�
�±��Ǽ��ּ��������Ĺ��ܡ���Ч�ɷ֡�
| �������� | ����� | ������Ư | Ư�� |
| �� �� | ��Ч����۹�������ζ | Ưϴ���ʹɫ�ʸ����� | ����Ư���������� |
| ��Ч�ɷ� | HCl | H2O2 | NaClO |
�ݱ��ش��������⣺
��1��������顱�ܷ�������������ã� ��
��2��������顱�����ڴ���ʯ���������� ����ʢ��������������Ư��Һ����Թ��У��������������̣��۲쵽�������� ��
��3�� ������顱�롰Ư�������ܻ��á�����������ײ���һ���ж����壬��ѧ����ʽΪ��NaClO + 2HCl =" NaCl" + X�� + H2O����X�Ļ�ѧʽΪ ��