��Ŀ����
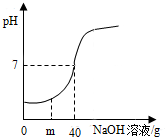 ʵ�������ⶨһƿ��ǩ�����ϡ�����������������ȡ20gϡ������Ʒ����10%��NaOH��Һ����������Ʒ�У��ӱ��ò��������裬����NaOH��Һ�IJ��ϼ��룬��Һ��pH������NaOH��Һ�����Ĺ�ϵ��ͼ��ʾ�����ʣ�
ʵ�������ⶨһƿ��ǩ�����ϡ�����������������ȡ20gϡ������Ʒ����10%��NaOH��Һ����������Ʒ�У��ӱ��ò��������裬����NaOH��Һ�IJ��ϼ��룬��Һ��pH������NaOH��Һ�����Ĺ�ϵ��ͼ��ʾ�����ʣ���1��NaOH��Һ�����ᷴӦ�Ļ�ѧ����ʽΪ��
��2��������NaOH��Һmgʱ����Һ�е�����Ϊ
��40g10%��KOH��Һ����ͬ�����ᷴӦ��������Һ��pH
��3����NaOH��Һ��ϡ����ǡ����ȫ��Ӧʱ��������Һ����������
��4������ϡ���������ʵ����������������ȷ��0.01%����
���㣺���ݻ�ѧ��Ӧ����ʽ�ļ���,�й��������������ļ���,��Һ���������pHֵ�Ĺ�ϵ
ר�⣺�ۺϼ��㣨ͼ���͡������͡��龰�ͼ����⣩
��������1������NaOH��Һ�����ᷴӦ�����Ȼ��ƺ�ˮ��д����Ӧ�Ļ�ѧ����ʽ��
��2��������Һ��pH��ϡ������NaOH��Һ�ķ�Ӧ������Һ�е����ʣ�����40g10%��KOH��Һ����ͬ�����ᷴӦ��������
��3�����������غ㶨�ɷ������
��4������ϡ������NaOH��Һ�ķ�Ӧ�ķ���ʽ����NaOH���������ϡ��������������������ϡ�������������������
��2��������Һ��pH��ϡ������NaOH��Һ�ķ�Ӧ������Һ�е����ʣ�����40g10%��KOH��Һ����ͬ�����ᷴӦ��������
��3�����������غ㶨�ɷ������
��4������ϡ������NaOH��Һ�ķ�Ӧ�ķ���ʽ����NaOH���������ϡ��������������������ϡ�������������������
����⣺
��1��NaOH��Һ�����ᷴӦ�����Ȼ��ƺ�ˮ����Ӧ�Ļ�ѧ����ʽΪ��NaOH+HCl�TNaCl+H2O��
��2����ͼʾ��֪������NaOH��Һmgʱ����Һ��pHС��7��˵�������NaOH��ȫ��Ӧ����ʣ���ϡ���ᣬ���ԣ���Һ�к��е�����Ϊ��NaCl��HCl��
��ͼʾ��֪������NaOH��Һ40gʱ����Һ��pH����7����40g10%��KOH��Һ����ͬ�����ᷴӦ��
NaOH+HCl�TNaCl+H2O KOH+HCl�TKCl+H2O
40 36.5 56 36.5
�ɴ˿�֪��40g10%��KOH��Һ����ȫ���ģ�������ʣ�࣬pHС��7��
��3����ͼʾ��֪������Һ��pH����7�����ĵ�NaOH��Һ������Ϊ40g��������Һ���������ǣ�20g+40g=60g��
��4��20gϡ������Ʒ�������ʵ�����Ϊx 40gNaOH��Һ�����ʵ����� 40g��10%=4g
NaOH+HCl�TNaCl+H2O
40 36.5
4g x
=
��ã�x=3.65g
ϡ�����������������Ϊ
��100%=18.25%
�𰸣�
��1��
NaOH+HCl�TNaCl+H2O
��2��NaCl��HCl��=
��3��60��
��4��ϡ���������ʵ���������18.25%
��1��NaOH��Һ�����ᷴӦ�����Ȼ��ƺ�ˮ����Ӧ�Ļ�ѧ����ʽΪ��NaOH+HCl�TNaCl+H2O��
��2����ͼʾ��֪������NaOH��Һmgʱ����Һ��pHС��7��˵�������NaOH��ȫ��Ӧ����ʣ���ϡ���ᣬ���ԣ���Һ�к��е�����Ϊ��NaCl��HCl��
��ͼʾ��֪������NaOH��Һ40gʱ����Һ��pH����7����40g10%��KOH��Һ����ͬ�����ᷴӦ��
NaOH+HCl�TNaCl+H2O KOH+HCl�TKCl+H2O
40 36.5 56 36.5
�ɴ˿�֪��40g10%��KOH��Һ����ȫ���ģ�������ʣ�࣬pHС��7��
��3����ͼʾ��֪������Һ��pH����7�����ĵ�NaOH��Һ������Ϊ40g��������Һ���������ǣ�20g+40g=60g��
��4��20gϡ������Ʒ�������ʵ�����Ϊx 40gNaOH��Һ�����ʵ����� 40g��10%=4g
NaOH+HCl�TNaCl+H2O
40 36.5
4g x
| 40 |
| 36.5 |
| 4g |
| x |
��ã�x=3.65g
ϡ�����������������Ϊ
| 3.65g |
| 20g |
�𰸣�
��1��
NaOH+HCl�TNaCl+H2O
��2��NaCl��HCl��=
��3��60��
��4��ϡ���������ʵ���������18.25%
������������һ�����ͼ��ļ����⣬����ʱ�ҵ�PH=7��ǡ�÷�Ӧ�㣬�����û�ѧ����ʽ�ļ�����н���ǽ����ͻ�ƿڣ�

��ϰ��ϵ�д�
 ����ѵ�����⿼ϵ�д�
����ѵ�����⿼ϵ�д�
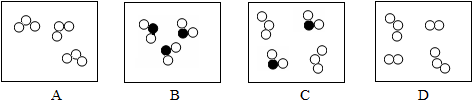
�����Ŀ
�������з�Ӧ��ʾ��ͼ�жϣ��÷�Ӧһ�����ڣ�������


| A���ֽⷴӦ | B���û���Ӧ |
| C�����Ϸ�Ӧ | D��������Ӧ |