��Ŀ����
��ȤС��ѡ�õ�15���е�Bװ�ã�װ�����������ã���CO2���������й�ʵ�飮
��1��ʵ������ȡCO2�Ļ�ѧ����ʽΪ______��
��2����ȤС����ʯ��ʯ����ε��������Ƶ����ᣬ������������ͨ�����ʯ��ˮ�У���ʯ��ˮʼ��û�л�������ʯ��ˮΪʲôû�л����أ��������ñ���ʵ����Ʒ����ѡ�Լ�����ѡ�Լ���Na2CO3��Һ���ữ��AgNO3��Һ����ɫʯ����Һ�����̽�����̣�
��������롿��______����______�����������ֲ���ͬʱ���ڣ�
�������ʵ�顿�������ʵ�鷽������֤����ٺͲ���ڣ�����������������±���
��1��ʵ������ȡCO2�Ļ�ѧ����ʽΪ______��
��2����ȤС����ʯ��ʯ����ε��������Ƶ����ᣬ������������ͨ�����ʯ��ˮ�У���ʯ��ˮʼ��û�л�������ʯ��ˮΪʲôû�л����أ��������ñ���ʵ����Ʒ����ѡ�Լ�����ѡ�Լ���Na2CO3��Һ���ữ��AgNO3��Һ����ɫʯ����Һ�����̽�����̣�
��������롿��______����______�����������ֲ���ͬʱ���ڣ�
�������ʵ�顿�������ʵ�鷽������֤����ٺͲ���ڣ�����������������±���
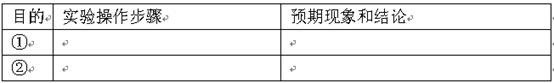
| Ŀ�� | ʵ��������� | Ԥ������ͽ��� |
| �� | ______ | ______ |
| �� | ______ | ______ |
��1����Ӧ����̼��ƺ��Ȼ��⣬���������Ȼ��ơ�ˮ��������̼���ù۲취��ƽ���ɣ�������̼��������������ţ�
��2��û�����˵�����ܱ��ʻ�����Ȼ��⣬��֤���������Ƿ���ʣ�����Һ���Ƿ����������ƣ����뺬̼������ӵĿ������Σ�����г�����˵��û���ʣ����û�г���˵���Ѿ����ʣ���֤����Ĵ��ڣ�������������Һ���а�ɫ����˵�������Ȼ��⣬�����У�
�ʴ�Ϊ����1��CaCO3+2HCl=CaCl2+H2O+CO2��
��2�������롿�ٳ���ʯ��ˮ�Ѿ�ʧЧ�����ʣ� ��CO2�л���HCl����֤��
��2��û�����˵�����ܱ��ʻ�����Ȼ��⣬��֤���������Ƿ���ʣ�����Һ���Ƿ����������ƣ����뺬̼������ӵĿ������Σ�����г�����˵��û���ʣ����û�г���˵���Ѿ����ʣ���֤����Ĵ��ڣ�������������Һ���а�ɫ����˵�������Ȼ��⣬�����У�
�ʴ�Ϊ����1��CaCO3+2HCl=CaCl2+H2O+CO2��
��2�������롿�ٳ���ʯ��ˮ�Ѿ�ʧЧ�����ʣ� ��CO2�л���HCl����֤��
| Ŀ�� | ʵ��������� | Ԥ������ͽ��� |
| �� | ��ȡ����ԭ����ʯ��ˮ���Թܣ�Ȼ��μ�����Na2CO3��Һ | ��������������ԭʯ��ˮ�Ѿ�ʧЧ���������ȷ������Һ����ǣ���ԭʯ��ˮû��ʧЧ������ٲ���ȷ |
| �� | ȡͨ����û�б���ǵ�ʯ��ˮ���Թܣ�Ȼ������ữ����������Һ����ȡ�ữ����������Һ���Թ��У�Ȼ��ͨ���Ƶõ����壩 | ����Һ�а�ɫ�������ɣ���CO2�л���HCl���������ȷ������Һû������������CO2��û����HCl������ڲ���ȷ |

��ϰ��ϵ�д�
 ������ϰ�ο����뵥Ԫ���ϵ�д�
������ϰ�ο����뵥Ԫ���ϵ�д�
�����Ŀ