��Ŀ����
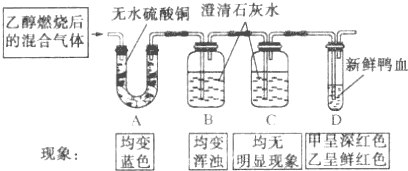 �ס���С��ͬѧΪ��ȷ���Ҵ���������ȼ�յIJ�����Ƿֱ��Ƚ��Ҵ��������е�ȼ������ȼ�պ�Ļ�������������ʵ�飮����ˮ����ͭ��ˮ�����ɫ������ѼѪ��O2�����ɫ����CO2�ʰ���ɫ����CO���ʺ�ɫ�����ס�����С���ʵ��˳�۲쵽��������ͼ��ʾ��
�ס���С��ͬѧΪ��ȷ���Ҵ���������ȼ�յIJ�����Ƿֱ��Ƚ��Ҵ��������е�ȼ������ȼ�պ�Ļ�������������ʵ�飮����ˮ����ͭ��ˮ�����ɫ������ѼѪ��O2�����ɫ����CO2�ʰ���ɫ����CO���ʺ�ɫ�����ס�����С���ʵ��˳�۲쵽��������ͼ��ʾ����������ʵ�飬�ش��������⣺
��1����С��ȷ���Ҵ�ȼ�յIJ��������
H2O��CO2
H2O��CO2
�� ��С��ȷ���Ҵ�ȼ�յIJ��������H2O��CO2��CO
H2O��CO2��CO
����2�������ס�������С��ȷ���IJ���Ϊʲô��ͬ��
����ʵ��ʱ�������㣬�Ҵ�����ȫȼ�գ�����ʵ��ʱ���������㣬�Ҵ�����ȫȼ��
����ʵ��ʱ�������㣬�Ҵ�����ȫȼ�գ�����ʵ��ʱ���������㣬�Ҵ�����ȫȼ��
����3����A��B����װ��˳��Ե�������Ϊʲô��
���У���Ϊװ�öԵ�����֤���Ҵ�ȼ�յIJ������Ƿ���ˮ
���У���Ϊװ�öԵ�����֤���Ҵ�ȼ�յIJ������Ƿ���ˮ
����4������̸̸����ʹ��ȼ�ϵ�һ�ַ�����
ʹȼ�ϳ��ȼ�գ������㹻��Ŀ�������������
ʹȼ�ϳ��ȼ�գ������㹻��Ŀ�������������
����5���Ҵ����׳�
�ƾ�
�ƾ�
����������
����
������������������������Դ�����Ļ�ѧʽΪC2H5OH
C2H5OH
����������ʵ�������ǣ��Ҵ�ȼ�պ�Ļ������ͨ��A��A����ɫ��֤��ȼ�ղ�������ˮ���ɣ�B����ǣ�֤��ȼ�ղ������ж�����̼���ɣ�C������������֤��������̼�ѱ�B�е��Լ������꣬D�Թ��У��׳����ɫ��˵��������ʵ��β������һ����̼���ҳ��ʺ�ɫ��˵��������ʵ��β������һ����̼��
����⣺��1��A��������˵������ˮ���ɣ�B������ǣ�˵�����ж�����̼���ɣ�D�Թ��м׳����ɫ��˵��������ʵ��β������һ����̼���ɣ��ҳ��ʺ�ɫ��˵��������ʵ��β������һ����̼���ɣ�
�ʴ�Ϊ��H2O��CO2��H2O��CO2��CO��
��2����Ϊ�IJ�����H2O��CO2���ҵIJ�����H2O��CO2��CO���������ֲ����֪������ȫȼ�գ��Ҳ���ȫȼ�գ�
�ʴ�Ϊ������ʵ��ʱ�������㣬�Ҵ�����ȫȼ�գ�����ʵ��ʱ���������㣬�Ҵ�����ȫȼ�գ�
��3������AB�Ե���������ȼ�պ�Ļ������ͨ��B����������ԭ���Ļ������������ˮ����ʹA��������Ϊ�����Bװ���г���ʱ�����ˮ��
�ʴ�Ϊ�����У���Ϊװ�öԵ�����֤���Ҵ�ȼ�յIJ������Ƿ���ˮ��
��4��Ϊ�˽�Լȼ�ϣ���ֹ��Ⱦ���������Ծ���ʹȼ�ϳ��ȼ�գ�
�ʴ�Ϊ��ʹȼ�ϳ��ȼ�գ������㹻��Ŀ���������������
��5���Ҵ����׳ƾƾ�������������Դ�����Ļ�ѧʽΪC2H5OH���ʴ�Ϊ���ƾ���������C2H5OH��
�ʴ�Ϊ��H2O��CO2��H2O��CO2��CO��
��2����Ϊ�IJ�����H2O��CO2���ҵIJ�����H2O��CO2��CO���������ֲ����֪������ȫȼ�գ��Ҳ���ȫȼ�գ�
�ʴ�Ϊ������ʵ��ʱ�������㣬�Ҵ�����ȫȼ�գ�����ʵ��ʱ���������㣬�Ҵ�����ȫȼ�գ�
��3������AB�Ե���������ȼ�պ�Ļ������ͨ��B����������ԭ���Ļ������������ˮ����ʹA��������Ϊ�����Bװ���г���ʱ�����ˮ��
�ʴ�Ϊ�����У���Ϊװ�öԵ�����֤���Ҵ�ȼ�յIJ������Ƿ���ˮ��
��4��Ϊ�˽�Լȼ�ϣ���ֹ��Ⱦ���������Ծ���ʹȼ�ϳ��ȼ�գ�
�ʴ�Ϊ��ʹȼ�ϳ��ȼ�գ������㹻��Ŀ���������������
��5���Ҵ����׳ƾƾ�������������Դ�����Ļ�ѧʽΪC2H5OH���ʴ�Ϊ���ƾ���������C2H5OH��
���������������ۺ�ʵ���⣬������Ĺؼ���Ū��ʵ�����̣��ѶȽϴ�

��ϰ��ϵ�д�
 �㽭��У��ʦ���ϵ�д�
�㽭��У��ʦ���ϵ�д�
�����Ŀ



