��Ŀ����
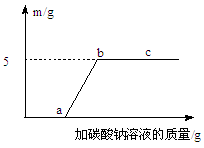
ijͬѧ��ʵ���ҷ���һƿ��̼���ƺ��Ȼ�����ɵĻ����Һ��Ϊ�˲ⶨ�û����Һ��̼���Ƶ�������������ͬѧ���������ʵ�飺ȡ�û����Һ50g����������μ���ϡ���ᣬ���������������Ϊ15g��30g��45g��60gʱ������������������±���������ܽ�Ⱥ��Բ��ƣ���
��1���ڢ�������nΪ g��
��2�������Һ��̼���Ƶ����������Ƕ��٣���д��������̣������ȷ��0.1%��
| | �ڢ��� | �ڢ��� | �ڢ��� | �ڢ��� |
| ϡ���������/g | 15 | 30 | 45 | 60 |
| �������������/g | 1.8 | n | 4.4 | 4.4 |
��2�������Һ��̼���Ƶ����������Ƕ��٣���д��������̣������ȷ��0.1%��
��1��3.6g;(2)21.2%
�����������1��15gϡ������ȫ��Ӧ���ɶ�����̼������Ϊ1.8g������30g������ȫ��Ӧ���ɶ�����̼����Ϊ3.6g��
��2������ǰ����ʵ�����ݷ�����֪ÿ15��������ȫ��Ӧ����1.8�˶�����̼����45��������ȫ��ӦӦ����5.4�˶�����̼���ڵ�����ʵ���м���45������ֻ����4.4�˶�����̼��˵��������ʵ����������ʣ�̼࣬���Ʒ�Ӧ�꣬��ȫ��Ӧ�������������Ϊ4.4�ˣ�������4.4g������̼����Ҫ�μӷ�Ӧ��̼���Ƶ�����Ϊx����
Na2CO3 +2HCl=2NaCl+CO2��+H2O
106 44
x 4.4g
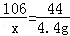
x=10.6g
���Һ��̼���Ƶ����������ǣ�
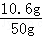 ��100%=21.2%
��100%=21.2%�𣺻��Һ��̼���Ƶ�����������21.2%��

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ