��Ŀ����
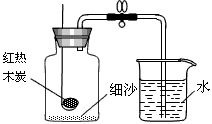
 С��Ϊ�ⶨ��������������������ͼװ�ý���ʵ�飮
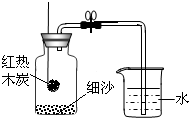
С��Ϊ�ⶨ��������������������ͼװ�ý���ʵ�飮С�����װ�������Ժ�ʢ����������ľ̿��ȼ�ճ�Ѹ�����뼯��ƿ�У��������������������ȵ�ľ̿Ϩ����ȴ�����º��ɼУ���δ���ֵ�������
���Է���δ���ֵ��������ԭ����
��С��������ƿ�е�ϸɳ��Ϊһ����Һ���ظ�����ʵ�飬�е�������д�������ָ���Һ���õĻ�ѧ����ʽ��
����ʵ��ɹ������뼯��ƿ�е�ˮԼռƿ�ڿռ��
��������1�����÷�Ӧ��ֻ��װ���ڵ�������Ӧ����������ѹǿ�ͨ����������װ����Һ���������������������������ʵ�飻����ľ̿ȼ�պ�����������������Ķ�����̼���壬��˵���װ���ڵ�ѹǿ�������䣬�ʲ����е�������ķ�����
��2��Ҫ��ⶨ���������������������ѡ�����Һ�����������ơ��������ơ��������ص���Һ����������̼�������յ����Ϳ��ԴﵽĿ�ģ�
��3���������������������Ϊ
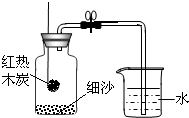
�����ʵ��ɹ������뼯��ƿ�е�ˮԼռƿ�ڿռ��
��
��2��Ҫ��ⶨ���������������������ѡ�����Һ�����������ơ��������ơ��������ص���Һ����������̼�������յ����Ϳ��ԴﵽĿ�ģ�
��3���������������������Ϊ
| 1 |
| 5 |
| 1 |
| 5 |
����⣺��1��ľ̿ȼ�������������������������µ����壬��ʹ����ƿ������ѹǿδ���٣�
��2������������Һ���������̼��Ӧ�����������ն�����̼���壬��Ӧ�Ļ�ѧ����ʽΪ2NaOH+CO2�TNa2CO3+H2O��
��3���������������������Ϊ
�����ʵ��ɹ������뼯��ƿ�е�ˮԼռƿ�ڿռ��
��
�ʴ�Ϊ����1��ľ̿ȼ�������������������������µ����壬��ʹ����ƿ������ѹǿδ����
��2��2NaOH+CO2�TNa2CO3+H2O
��3��
��
��2������������Һ���������̼��Ӧ�����������ն�����̼���壬��Ӧ�Ļ�ѧ����ʽΪ2NaOH+CO2�TNa2CO3+H2O��
��3���������������������Ϊ
| 1 |
| 5 |
| 1 |
| 5 |
�ʴ�Ϊ����1��ľ̿ȼ�������������������������µ����壬��ʹ����ƿ������ѹǿδ����
��2��2NaOH+CO2�TNa2CO3+H2O
��3��
| 1 |
| 5 |
| 1 |
| 5 |
���������⿼��ⶨ�����������������ҩƷѡ���������Լ�ʵ�鷽�������ۣ�����������ʱҪ��Ϥʵ��ԭ�������ܶ�ʵ�����ʧ�ܵ�ԭ��ӷ�Ӧ���ѡ���������״̬�����������������������ȷ��ѡ��

��ϰ��ϵ�д�
 Сѧ������ҵϵ�д�
Сѧ������ҵϵ�д� ��ʿһ��ȫͨϵ�д�
��ʿһ��ȫͨϵ�д�
�����Ŀ
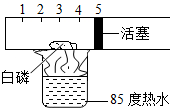 ij��ѧ��Ȥ�С��Ϊ�ⶨ�����������ĺ�������������̽��ʵ�飺
ij��ѧ��Ȥ�С��Ϊ�ⶨ�����������ĺ�������������̽��ʵ�飺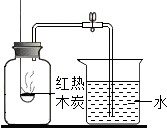
 С��Ϊ�ⶨ��������������������ͼװ�ý���ʵ�飮
С��Ϊ�ⶨ��������������������ͼװ�ý���ʵ�飮