��Ŀ����
ijУ��ѧʵ����ϣ�ͬѧ���û�ѧ����̽��һ�ָ���Ʒ�����ĺ�����ͬѧ�ǽ�11.4 g����Ʒ��20.0 gϡ���ᣨ��������������Ϊ120.0 g�ձ��С��ڻ�ѧ��Ӧ�����ж��ձ������е�ʣ����������Ĵγ�������¼���±���
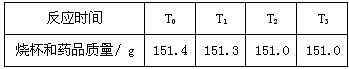
�����������һλС����
��1����Ӧ�в��������������� g��
��2���ֵ����ͷ�Ϊ��
��1����Ӧ�в��������������� g��
��2���ֵ����ͷ�Ϊ��

����������ձ��еĺ�ɫ����ȫ����̼���ʣ���ͨ�������ж����ָ����������͵ĸ֣�
��3��ij�ֳ���ұ��1000t���ֲָģ��躬������80%�ij�������ٶ֣�
��3��ij�ֳ���ұ��1000t���ֲָģ��躬������80%�ij�������ٶ֣�
��1��0.4
��2���⣺������к���������Ϊx
Fe + 2HCl===FeCl2 + H2 ��
56 2
x 0.4 g
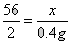 x=11.2 g
x=11.2 g
������̼��=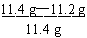 ��100% = 1.8% �����ڸ�̼��
��100% = 1.8% �����ڸ�̼��
��3��1000 t��(1-1.8%)��70%��80%=1753.6 t ��
1000t��(1-1.75%)��70%��80%=1754.5 t
����Ҫ���ֳ�����ʯ1753.6 t��
��2���⣺������к���������Ϊx
Fe + 2HCl===FeCl2 + H2 ��
56 2
x 0.4 g
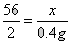 x=11.2 g
x=11.2 g ������̼��=
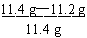 ��100% = 1.8% �����ڸ�̼��
��100% = 1.8% �����ڸ�̼����3��1000 t��(1-1.8%)��70%��80%=1753.6 t ��
1000t��(1-1.75%)��70%��80%=1754.5 t
����Ҫ���ֳ�����ʯ1753.6 t��

��ϰ��ϵ�д�
�����Ŀ
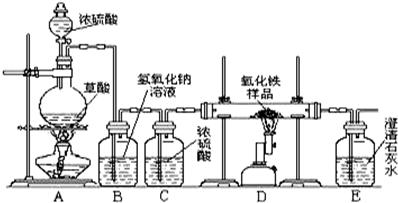
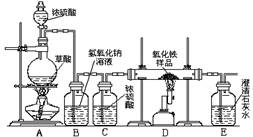
 ijУ��ѧ������ȤС���ͬѧ���о���ѧϰ����չʾ��һ������ͼ��ʾʵ��װ�ã�����ÿ����ѧ��Ӧ����ȫ����������Ʒ�е����ʲ��μӷ�Ӧ�����������ϣ�������Ũ�������ʱ���ȷ������·�Ӧ��H2C2O4
ijУ��ѧ������ȤС���ͬѧ���о���ѧϰ����չʾ��һ������ͼ��ʾʵ��װ�ã�����ÿ����ѧ��Ӧ����ȫ����������Ʒ�е����ʲ��μӷ�Ӧ�����������ϣ�������Ũ�������ʱ���ȷ������·�Ӧ��H2C2O4

 CO��+CO2��+H2O
CO��+CO2��+H2O