��Ŀ����
ˮ����Һ��ʥˮ������������ۣ�λ�����з����Ϊʡ���ص����ﱣ����λ�����С��й���ʤ�ǵ䡷֮�У�����Ⱥɽ��ب��������ģ�������䣬����С����֮�������Թž���ɽ��ˮ���������ǣ���˵����ȫ���ɵĴ�ʼ���������������ڴ˴�������ʥˮ��ϵ��Ȼʯ��������������ŭ�ţ�������ף������ף��ǽ���ɽ�������Ĵ��ڶ���ͷ��ʯ���ϣ�����Ȫ��������������ζ�ʣ�����Ϊ��ʥˮ����

��1���ݲⶨ����ʥˮ���к��������ȡ�п�������ơ��Ƶȶ��ֶ����������Ԫ�أ���ˡ�ʥˮ�����ڣ����������______��Ҫ���顰ʥˮ���Ƿ���Ӳˮ�IJ��������ǣ�______��
��2����ͼ2Ϊ����ԭ�ӵĺ���������Ϊ______���ڻ�ѧ��Ӧ����ԭ����______����õ�����ʧȥ�������ӣ�
��3����1977�꿪ʼ����ɽ�Ƴ��ô�ˮ��ɡ�ʥȪ���������˾ƴ���Ũ������䣸�ˬ����ʥȪ�����������ʵĻ�ѧʽ��______��
��4��ij��������A���ܽ��������ͼ��ʾ��
�ٴ�ͼ3���Եó�����A���ܽ�ȱ仯������______��
��С������ͼ3��70��ʱ��������ͼ4ʵ��С���۲쵽�������ӹ���ȫ���ܽ⣬������������______��30��ʱ
������Һ����������������______�����������0.1%����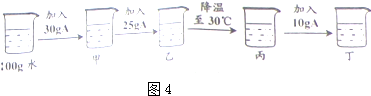

��1���ݲⶨ����ʥˮ���к��������ȡ�п�������ơ��Ƶȶ��ֶ����������Ԫ�أ���ˡ�ʥˮ�����ڣ����������______��Ҫ���顰ʥˮ���Ƿ���Ӳˮ�IJ��������ǣ�______��
��2����ͼ2Ϊ����ԭ�ӵĺ���������Ϊ______���ڻ�ѧ��Ӧ����ԭ����______����õ�����ʧȥ�������ӣ�
��3����1977�꿪ʼ����ɽ�Ƴ��ô�ˮ��ɡ�ʥȪ���������˾ƴ���Ũ������䣸�ˬ����ʥȪ�����������ʵĻ�ѧʽ��______��
��4��ij��������A���ܽ��������ͼ��ʾ��
�ٴ�ͼ3���Եó�����A���ܽ�ȱ仯������______��
��С������ͼ3��70��ʱ��������ͼ4ʵ��С���۲쵽�������ӹ���ȫ���ܽ⣬������������______��30��ʱ
������Һ����������������______�����������0.1%����

��1����ʥˮ���к��������ȡ�п�������ơ��Ƶȶ��ֶ����������Ԫ�أ��ɼ���ʥˮ�����ڻ���Ӳˮ�Ǻ��н϶�����Եĸơ�þ�������ˮ����ˮ��ָ�������ٺ������Ըơ�þ�������ˮ����ˮ�зֱ��������ķ���ˮ����ĭ�ḻ��Ϊ��ˮ����ĭ���١������϶��ΪӲˮ��
��2����ԭ�ӽṹʾ��ͼ��ԲȦ�ڵ�����Ϊ����������֪��Ԫ��ԭ�Ӻ�����34�����ӣ�����������Ϊ6����Ԫ�ص�ԭ���ڻ�ѧ��Ӧ��һ��Ҫ�õ�2�����Ӷ��γ��ȶ��ṹ��
��3���ƾ��Ļ�ѧʽΪC2H5OH��
��4������ͼ�п�֪������A���ܽ�����¶ȵ����߶���С�������ܽ�����߿�֪��70��ʱ100��ˮ��������52��A���ʣ��ɼ����ձ�������A������ȫ�ܽ⣬����30��ʱ���ܽ��Ϊ69�ˣ�����ȫ���ܽ�����65��A��
��100%=39.4%��
�ʴ�Ϊ����1��������ʢ��ʥˮ���Թ��У����������ķ���ˮ����������������ݽ��١������϶࣬˵����Ӳˮ����֮����ˮ��
��2��34���õ�
��3��C2H5OH
��4�������¶ȵ����߶���С���ڹ��岿���ܽ⣻39.4%
��2����ԭ�ӽṹʾ��ͼ��ԲȦ�ڵ�����Ϊ����������֪��Ԫ��ԭ�Ӻ�����34�����ӣ�����������Ϊ6����Ԫ�ص�ԭ���ڻ�ѧ��Ӧ��һ��Ҫ�õ�2�����Ӷ��γ��ȶ��ṹ��
��3���ƾ��Ļ�ѧʽΪC2H5OH��
��4������ͼ�п�֪������A���ܽ�����¶ȵ����߶���С�������ܽ�����߿�֪��70��ʱ100��ˮ��������52��A���ʣ��ɼ����ձ�������A������ȫ�ܽ⣬����30��ʱ���ܽ��Ϊ69�ˣ�����ȫ���ܽ�����65��A��
| 65g |
| 165g |
�ʴ�Ϊ����1��������ʢ��ʥˮ���Թ��У����������ķ���ˮ����������������ݽ��١������϶࣬˵����Ӳˮ����֮����ˮ��
��2��34���õ�
��3��C2H5OH
��4�������¶ȵ����߶���С���ڹ��岿���ܽ⣻39.4%

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ