��Ŀ����
���������������벻��ˮ����ͼ������ˮ����ˮ����ʾ��ͼ�������ʾ��ͼ�ش��������⣺
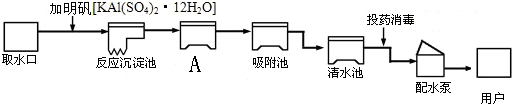
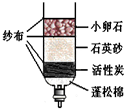
��1������ˮ��Al3+�����к������������������ˮ��Al3+����IJ�����______����ˮ���е�ˮ����______����������������
��2��A����ͨ��______����������ƣ���ȥˮ�еĹ������ʣ�
��3�����������������ƣ�NaClO2����Ӧ��ȡClO2�ķ�Ӧ����ʽΪ��Cl2+2NaClO2�T2ClO2+2X��X�Ļ�ѧʽΪ______��
��4����Ȼ������Щ��ˮ���Ⱥ�����һЩ��������ͨ����˵��ˮ����˵���˾�ˮ����ΪӲˮ����˾�ˮ�к���______�������ӷ��ţ���
��5��Ϊ��ʵ�����Ҫ��������ˮ��һ������Ϊ����ˮ����ʵ������ȡ����ˮ��������ƿ�ͨ��Ҫ���뼸����ʯ�����Ƭ��������______��

��1������ˮ��Al3+�����к������������������ˮ��Al3+����IJ�����______����ˮ���е�ˮ����______����������������
��2��A����ͨ��______����������ƣ���ȥˮ�еĹ������ʣ�
��3�����������������ƣ�NaClO2����Ӧ��ȡClO2�ķ�Ӧ����ʽΪ��Cl2+2NaClO2�T2ClO2+2X��X�Ļ�ѧʽΪ______��
��4����Ȼ������Щ��ˮ���Ⱥ�����һЩ��������ͨ����˵��ˮ����˵���˾�ˮ����ΪӲˮ����˾�ˮ�к���______�������ӷ��ţ���
��5��Ϊ��ʵ�����Ҫ��������ˮ��һ������Ϊ����ˮ����ʵ������ȡ����ˮ��������ƿ�ͨ��Ҫ���뼸����ʯ�����Ƭ��������______��
��1����������������к�����Ԫ�أ��������ˮ��Al3+����IJ��������Ǽ���������������ˮ���е�ˮ���п����Ե����ʣ����ڻ���
��2��ͨ�����˳�ȥˮ�еĹ������ʣ�
��3����Cl2+2NaClO2�T2ClO2+2X����ʽ������У���ԭ�ӡ���ԭ�ӡ���ԭ�ӵ���Ŀ�ֱ���4��2��2�����ұ�����ԭ�ӡ���ԭ�ӵ���Ŀ�ֱ���2��2���������غ㶨�ɷ�Ӧǰ��ԭ�ӵ����༰��Ŀ�����֪��X�Ļ�ѧʽ��NaCl��
��4������Ӳˮ�Ǻ��н϶�ơ�þ���ӻ������ˮ�����ԣ���˾�ˮ�к���Ca2+��Mg2+��
��5������ʱ��ͨ��Ҫ���뼸����ʯ�����Ƭ�������Ƿ�ֹ���У�
�ʴ�Ϊ����1��������������2�����ˣ���3��NaCl����4��Ca2+��Mg2+����5����ֹ���У�
��2��ͨ�����˳�ȥˮ�еĹ������ʣ�
��3����Cl2+2NaClO2�T2ClO2+2X����ʽ������У���ԭ�ӡ���ԭ�ӡ���ԭ�ӵ���Ŀ�ֱ���4��2��2�����ұ�����ԭ�ӡ���ԭ�ӵ���Ŀ�ֱ���2��2���������غ㶨�ɷ�Ӧǰ��ԭ�ӵ����༰��Ŀ�����֪��X�Ļ�ѧʽ��NaCl��
��4������Ӳˮ�Ǻ��н϶�ơ�þ���ӻ������ˮ�����ԣ���˾�ˮ�к���Ca2+��Mg2+��
��5������ʱ��ͨ��Ҫ���뼸����ʯ�����Ƭ�������Ƿ�ֹ���У�
�ʴ�Ϊ����1��������������2�����ˣ���3��NaCl����4��Ca2+��Mg2+����5����ֹ���У�

��ϰ��ϵ�д�
 ��ʦ�㲦��ϵ�д�
��ʦ�㲦��ϵ�д�
�����Ŀ