��Ŀ����
ʳ�����ճ�����ı���Ʒ��Ҳ����Ҫ�Ļ���ԭ�ϣ�һͬѧ��ij�ִ��ν����ᴿʵ�飬������ͼ��ʾ��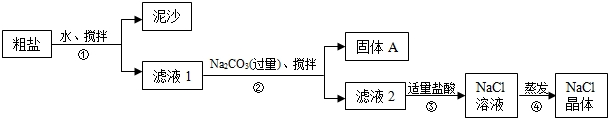
��ش�
��1������ٺ͢ڵIJ���������
��2���������Ϊ�õ������Ĺ���A���轫����A��ˮϴ�Ӹɾ��������ʵ��������A�Ƿ�ϴ��
��3���������֤��
| ���� | ��֤���� | ʵ������ | ���� |
| ����I������A�к���CaCO3 | ȡ��������A���Թ��У��μ�ϡ���ᣬ����Ϳ�г���ʯ��ˮ��С�ձ������Թܿ� | ___________ | ����I���� |
| ����II������A���BaCO3 |
ȡ��������A���Թ��У��ȵ���_________���ٵ���ϡNa2SO4��Һ | �����ݷų����ް�ɫ���� | ________ |
| ����III������Ƶõ�NaCl�����л�����Na2SO4 |
ȡ����NaCl�������Թ��У�������������ˮ�ܽ⣬��_______ | ________ | ����III���� |
��������1���ӿ��ǹ�����Һ��������֣�
��2���������Ƿ����ûϴ����̼���ƣ���Ҫ�ӳ�����̼���ƵIJ�ͬ���ʿ��ǣ�
��3���Ӽ���̼������ӵķ������֣�����ʵ�������Ƴ�ʵ�鷽���ͽ��ۣ��ж��Ƿ������������Ӿ��У�
��4�����ݶ�����̼���������̼��Ƶ��������������Ԫ�ص�������������Ʒ�����������ɣ�
��2���������Ƿ����ûϴ����̼���ƣ���Ҫ�ӳ�����̼���ƵIJ�ͬ���ʿ��ǣ�
��3���Ӽ���̼������ӵķ������֣�����ʵ�������Ƴ�ʵ�鷽���ͽ��ۣ��ж��Ƿ������������Ӿ��У�
��4�����ݶ�����̼���������̼��Ƶ��������������Ԫ�ص�������������Ʒ�����������ɣ�
����⣺��1���Ѳ�����Һ��Ĺ����Һ�����ķ������ǹ��ˣ�
��2������A����ʹ���ָʾ����ɫ����̼�����Լ��ԣ��ܹ�ʹ��ɫ��̪��죬���Եμ���ɫ��̪��������ɫ�仯���жϣ�
��3��̼������ӵļ��鷽�����ǵμ�ϡ���ᣬ��������ܹ�ʹ����ʯ��ˮ����ǵ����壬˵����̼������ӣ����������������ݲ�����ʯ��ˮ����ǣ����������֪û�б����ӣ�Ӧ���ȼ������ϡ���ᣬ�ṩ�����������ټ������û�г���˵��û�б����ӣ����ڶ��ֲ�������������Ӧ�ü������ᱵ��ϡ���ᣬ����г�������˵���������ƣ�
��4�����ݶ�����̼���������̼��Ƶ�����Ϊ0.3g�����Ԫ�ص�������0.3g��40%=0.12g�����ԼӸ�ʳ���и�Ԫ�ص���������=
��100%=1.2%��
�ʴ�Ϊ����1�����ˣ�
��2������������A�����Թ��ڣ����Թ��ڵ�������ˮ�������Թ��ڵμ���ɫ��̪��Һ�������̪��Һ��죬˵��ûϴ�ɾ����������죬��֤���Ѿ�ϴ�ɾ��ˣ�
��3�������������ݲ�����ʯ��ˮ����ǣ���֤�����Ǽ��������ϡ���ᣬ�����ǹ���A�в�����̼�ᱵ����֤�����Ǽ������ᱵ��ϡ���ᣬʵ���������а�ɫ�������ɣ�
��4��1.2%��
��2������A����ʹ���ָʾ����ɫ����̼�����Լ��ԣ��ܹ�ʹ��ɫ��̪��죬���Եμ���ɫ��̪��������ɫ�仯���жϣ�
��3��̼������ӵļ��鷽�����ǵμ�ϡ���ᣬ��������ܹ�ʹ����ʯ��ˮ����ǵ����壬˵����̼������ӣ����������������ݲ�����ʯ��ˮ����ǣ����������֪û�б����ӣ�Ӧ���ȼ������ϡ���ᣬ�ṩ�����������ټ������û�г���˵��û�б����ӣ����ڶ��ֲ�������������Ӧ�ü������ᱵ��ϡ���ᣬ����г�������˵���������ƣ�
��4�����ݶ�����̼���������̼��Ƶ�����Ϊ0.3g�����Ԫ�ص�������0.3g��40%=0.12g�����ԼӸ�ʳ���и�Ԫ�ص���������=
| 0.12g |
| 10g |
�ʴ�Ϊ����1�����ˣ�
��2������������A�����Թ��ڣ����Թ��ڵ�������ˮ�������Թ��ڵμ���ɫ��̪��Һ�������̪��Һ��죬˵��ûϴ�ɾ����������죬��֤���Ѿ�ϴ�ɾ��ˣ�
��3�������������ݲ�����ʯ��ˮ����ǣ���֤�����Ǽ��������ϡ���ᣬ�����ǹ���A�в�����̼�ᱵ����֤�����Ǽ������ᱵ��ϡ���ᣬʵ���������а�ɫ�������ɣ�
��4��1.2%��
���������벻����Һ��Ĺ�����Һ���ù��˵ķ���������ʵ�鷽�����Ƴ�ʵ������ͽ��ۣ�����ʵ������Ҳ�ɷ��Ƴ�ʵ�鷽���ͽ��ۣ�

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ