��Ŀ����
����ͭ�Ǿ���ͭ����Ҫԭ�ϣ�ij��������ͭ��CuS��������ͭ��ѡ����������
����һ��CuS����˾����������������¸������е�O2������Ӧ����CuSO4
��������2CuS+3O2![]() 2CuO+2SO2 ��CuO+H2SO4= CuSO4 +H2O
2CuO+2SO2 ��CuO+H2SO4= CuSO4 +H2O
��1����д������һ��Ӧ�Ļ�ѧ����ʽ ��
��2���ӻ��������ĽǶȿ��ǣ�����������Ӧѡ �������������ǣ�����д�����㣩
�� ��
��1�� CuS+2O2![]() CuSO4
CuSO4
��2�� ����һ �ٲ����ȣ���Լ��Դ ��û�и���Ʒ��û����Ⱦ���������ʲ������������ɫ��ѧҪ��

��ϰ��ϵ�д�
�����Ŀ
 ��
��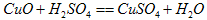
 2CuO+2SO2��CuO+H2SO4=CuSO4+H2O
2CuO+2SO2��CuO+H2SO4=CuSO4+H2O