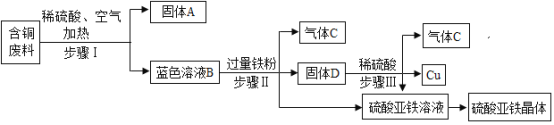
��Ŀ����
����Ŀ��ʵ���ҳ��õĸ��������ʯ������CaO����NaOH�Ļ����,�����������ˮ������CO2��Ӧ������ijͬѧ��һƿ���õ�����ʯ������������̽��������֪��Ca(OH)2+Na2CO3=CaCO3��+2NaOH��
��1�����룺
��������û�б���,����ʯ����ֻ����CaO������NaOH��
������������ȫ����,����ʯ����ȫ�������CaCO3��Na2CO3��
��CaO��ˮ��Ӧ�ķ�Ӧ����ʽΪ_________��
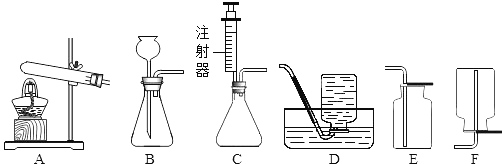
��2��ʵ�飺��ͼ��ʾ��
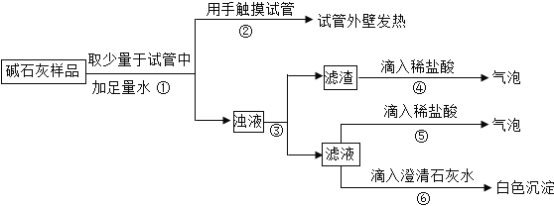
��3���жϣ�
a��Ca(OH)��CaCO3��Na2CO3Ͷ�뵽ˮ�в������,���ɲ����������жϣ�������______�����������������
b NaOH��¶�ڿ����б��ʵĻ�ѧ����ʽΪ______���ɲ����ݢ������жϣ���Һ�к���______��д��ѧʽ�����ɴ��жϲ�����______�������������������
c�ۺ�a��b�Ľ���,�жϸ���Ʒ�������Ϊ______��
��4����չ��������ʵ��˵��,ʵ����������ʯ����Ӧ______���棻�������в�������������______
���𰸡�CaO+H2O=Ca(OH)2 ������ 2NaOH+CO2=Na2CO3+H2O Na2CO3 ������ ���ֱ��� �ܷ� ����
��������
��1�����룺CaO��ˮ��Ӧ�����������ƣ���Ӧ�Ļ�ѧ����ʽΪ��CaO+H2O=Ca(OH)2�����CaO+H2O=Ca(OH)2��
��3���жϣ�a���ڲ����ڵ������Ƿ��ȣ���ô�����������ƺ�ˮ��Ӧ���ȣ���NaOH����ˮ���ȵ��µģ�˵����Ʒ�к��������ƣ����������ƣ��������ƺ��������ƣ�˵����Ʒû����ȫ���ʣ��������������������������
b NaOH��¶�ڿ�����������еĶ�����̼��Ӧ������̼���ƺ�ˮ����˱��ʵĻ�ѧ����ʽΪ2NaOH+CO2=Na2CO3+H2O���ɲ����ݣ�����ϡ���������ݲ�����˵�����溬�п����Ե�̼���Σ��������֪��Һ�к���̼���ƣ�����Һ�к��е�̼���ƺ�ʯ��ˮ��Ӧ���ɲ�����ˮ��̼��ƺ�ˮ������֤������Һ���溬��̼������Һ��֤���˲����������������2NaOH+CO2=Na2CO3+H2O��Na2CO3����������
c��a�ó��Ľ��ۣ�ȫ�������Ǵ���ģ���b�ó��Ľ����ǣ�û�б����Ǵ���ģ��ۺ�a��b�Ľ��ۣ��жϸ���Ʒ�������Ϊ���ֱ��ʣ����ʵ�ԭ�����������ƺͿ����еĶ�����̼��Ӧ����̼���ƺ�ˮ��������ֱ��ʡ�
��4����ʯ���ڿ����м��ױ��ʣ�������Ҫ�ܷⱣ�棬����ʱ��Һ������⽦����Ҫ������������������ܷ⣻������

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�