��Ŀ����
����A��B��C���ֳ��г������ʣ���������Ϊ���������� ���������ش����⡣
���������ش����⡣
��1����A�м���C ���ʣ�������B��BΪһ�ּA��B������ͬ�Ľ���Ԫ�أ���CΪ____________����A���뱥�͵�B��Һ�У����ְ�ɫ���ǣ��ð�ɫ������Ϊ_________��������Ӧ�Ļ�ѧ����ʽΪ_______________________��
��2����A������Ԫ�أ�A�������ֽⷴӦͬʱ����B��C����A�Ļ�ѧʽΪ_______,�÷�Ӧ�Ļ�ѧ����ʽΪ____________________________________________��
��3����A��һ�ֺ�ɫ���壬������ϡ���ᣬ�õ���ɫ��Һ����A��B��C�������ʴ����������ϵA��B��C����C������������������______________��
A���� B���� C���� D�������� E������
��1��H2O��ˮ CaO + H2O == Ca(OH)2 Ca(OH)2
��2��HCl CaCO3 + 2HCl ==== CaCl 2 + H2O + CO2�������������𰸸�����
��3��A B C D E ���˿�Ϊ2��,���3������0�������3——4����1����

��ϰ��ϵ�д�
�����Ŀ
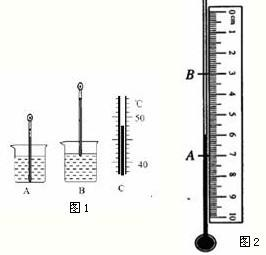 �ڡ��۲�ˮ�ķ��ڡ�ʵ���У�
�ڡ��۲�ˮ�ķ��ڡ�ʵ���У���1��ͼ1��С��ͬѧ���¶ȼƲ�С�ձ���ˮ�ij���ʱ�IJ���ͼ��A�Dz������̣�B�Ƕ������̣�C�Ƕ�ȡ���¶ȣ�
��Aͼ�в����Ĵ�����
��Bͼ�ж����Ĵ�����
����������ȷ������Cͼ��֪��ʱ�ձ���ˮ��ʵ���¶���
��2����ʵ���������ͬѧ���ˮ�ķе�ֱ�Ϊ97�桢93�桢102�棻��ͬѧ���뵼�����������ԭ���Ǹ����õ��¶ȼ���ƫ��������һ���ķ�����֤����룺
��3��
| ���� | ���̵� | �е� |
| ˮ�� | -39�� | 357�� |
| �ƾ� | -117�� | 78�� |
�ڡ��۲�ˮ�ķ��ڡ�ʵ���У�
��1��ͼ1��С��ͬѧ���¶ȼƲ�С�ձ���ˮ�ij���ʱ�IJ���ͼ��A�Dz������̣�B�Ƕ������̣�C�Ƕ�ȡ���¶ȣ�
��Aͼ�в����Ĵ�����______��
��Bͼ�ж����Ĵ�����______��
����������ȷ������Cͼ��֪��ʱ�ձ���ˮ��ʵ���¶���______�森
��2����ʵ���������ͬѧ���ˮ�ķе�ֱ�Ϊ97�桢93�桢102�棻��ͬѧ���뵼�����������ԭ���Ǹ����õ��¶ȼ���ƫ��������һ���ķ�����֤����룺______��
��3��
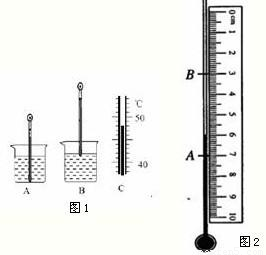
С��Ҫ����һ֧�ܲ�ˮ�е���¶ȼƣ����б������е��������ʣ���Ӧѡ���ұ��е�______���������ʣ�ԭ����______����һ������ѹ�£����¶ȼ��Ⱥ�����ˮ�����ͷ�ˮ�У��ֱ����¶ȼ���Һ���ﵽ��λ��A��B��Ȼ���¶ȼƷ��ڿ̶ȳ��ԣ���ͼ2��ʾ��ͼ�п̶ȳߵķֶ�ֵ��______�����ʱ�¶ȼ���ʾ���¶���______��
��1��ͼ1��С��ͬѧ���¶ȼƲ�С�ձ���ˮ�ij���ʱ�IJ���ͼ��A�Dz������̣�B�Ƕ������̣�C�Ƕ�ȡ���¶ȣ�
��Aͼ�в����Ĵ�����______��
��Bͼ�ж����Ĵ�����______��
����������ȷ������Cͼ��֪��ʱ�ձ���ˮ��ʵ���¶���______�森
��2����ʵ���������ͬѧ���ˮ�ķе�ֱ�Ϊ97�桢93�桢102�棻��ͬѧ���뵼�����������ԭ���Ǹ����õ��¶ȼ���ƫ��������һ���ķ�����֤����룺______��
��3��
| ���� | ���̵� | �е� |
| ˮ�� | -39�� | 357�� |
| �ƾ� | -117�� | 78�� |
