��Ŀ����
��2004?��ɽ��С����ⶨCu-Zn�Ͻ�Cu-Ag�Ͻ���ͭ������������ʵ����ֻ�ṩһƿδ��������������ϡ����ͱ�Ҫ����������1������Ϊ�ܲ����ͭ��������������______�Ͻ�
��2��С��ȡ�úϽ�ķ�ĩ32.5g���������������ַ�Ӧ�����ⶨ��������0.4g����������úϽ���ͭ������������
��3�����������������������������Ϊʵ��ʱ�����ṩ�Ͳ����������______��ѡ����ţ���
A���μӷ�Ӧ�ĺϽ�����
B���μӷ�Ӧ��ϡ���������
C���μӷ�Ӧ��ϡ�����������ܶ�
D�����������������
���𰸡���������1���������ṩ��ҩƷ��֪������Ҫ����ĺϽ��е����ֽ�������һ������֮ǰ��һ������֮��
��2��������ǰ����������ķ�Ӧ�������п�ͽ���ͭ����������������Ͻ���ͭ������������
��3��Ҫ������������ʵ�����������������������ʺ���Һ������������Һ��������Ҫ��������ʵ�������Ҫ���������������������������
����⣺��1��ͭ�Ļ�Ա�����������ϡ�ᷢ����Ӧ��п�Ļ�Ա���ǿ������ϡ�ᷢ����Ӧ�����ͭп�Ͻ��ܲⶨ��ͭ�����������������Ļ�Ա�������Ҳ����ϡ�ᷢ����Ӧ�����ͭ���Ͻ��ܲⶨ��ͭ������������
��2���裺�Ͻ���п������Ϊx
Zn+2HCl=ZnCl2+H2��
65 2
x 0.4g
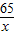 =
=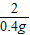
x=13g
�Ͻ���ͭ������Ϊ��32.5g-13g=19.5g
�Ͻ���ͭ����������= ×100%=60%
×100%=60%
�𣺺Ͻ���ͭ����������Ϊ60%��
��3��Ҫ�������������������������֪�����������ʵ���������Һ���������μӷ�Ӧ�����������ʵ�����������������������ͨ����ѧ����ʽ�������Ȼ�����ʵ�����÷ϡ�����������×100%��Ϊ��������������������μӷ�Ӧ�ĺϽ�������ǻ��������������м���ʱ����ֱ��ʹ�ã�ʵ��ʱ�������μӷ�Ӧ��ϡ�����������������������������ѡBC��CD��
�ʴ�Ϊ��
��1��Cu-Zn��
��2���裺�Ͻ���п������Ϊx
Zn+2HCl=ZnCl2+H2��
65 2
x 0.4g
 =
=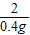
x=13g
�Ͻ���ͭ������Ϊ��32.5g-13g=19.5g
�Ͻ���ͭ����������=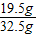 ×100%=60%
×100%=60%
�𣺺Ͻ���ͭ����������Ϊ60%��
��3��B D����C D����
���������⿼��������˳����Ӧ�ã�ͬʱ������ݻ�ѧ����ʽ�ļ��㣬���Ӳ�ͬ�Ƕȿ���ѧ�����������������
��2��������ǰ����������ķ�Ӧ�������п�ͽ���ͭ����������������Ͻ���ͭ������������
��3��Ҫ������������ʵ�����������������������ʺ���Һ������������Һ��������Ҫ��������ʵ�������Ҫ���������������������������
����⣺��1��ͭ�Ļ�Ա�����������ϡ�ᷢ����Ӧ��п�Ļ�Ա���ǿ������ϡ�ᷢ����Ӧ�����ͭп�Ͻ��ܲⶨ��ͭ�����������������Ļ�Ա�������Ҳ����ϡ�ᷢ����Ӧ�����ͭ���Ͻ��ܲⶨ��ͭ������������
��2���裺�Ͻ���п������Ϊx
Zn+2HCl=ZnCl2+H2��
65 2
x 0.4g
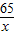 =
=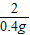
x=13g
�Ͻ���ͭ������Ϊ��32.5g-13g=19.5g
�Ͻ���ͭ����������=
 ×100%=60%
×100%=60% �𣺺Ͻ���ͭ����������Ϊ60%��
��3��Ҫ�������������������������֪�����������ʵ���������Һ���������μӷ�Ӧ�����������ʵ�����������������������ͨ����ѧ����ʽ�������Ȼ�����ʵ�����÷ϡ�����������×100%��Ϊ��������������������μӷ�Ӧ�ĺϽ�������ǻ��������������м���ʱ����ֱ��ʹ�ã�ʵ��ʱ�������μӷ�Ӧ��ϡ�����������������������������ѡBC��CD��
�ʴ�Ϊ��
��1��Cu-Zn��
��2���裺�Ͻ���п������Ϊx
Zn+2HCl=ZnCl2+H2��
65 2
x 0.4g
 =
=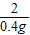
x=13g
�Ͻ���ͭ������Ϊ��32.5g-13g=19.5g
�Ͻ���ͭ����������=
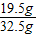 ×100%=60%
×100%=60% �𣺺Ͻ���ͭ����������Ϊ60%��
��3��B D����C D����
���������⿼��������˳����Ӧ�ã�ͬʱ������ݻ�ѧ����ʽ�ļ��㣬���Ӳ�ͬ�Ƕȿ���ѧ�����������������

��ϰ��ϵ�д�
�����Ŀ