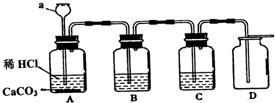
��Ŀ����
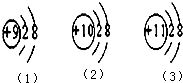 �ش��������⣺
�ش��������⣺��1�����ʵķ�����ж��֣��������������������ˮ��H2O����������O2����������̼��CO2�������飨CH4�����������е����ְ�����һ�ֱ��ڣ�����һ�ֲ������ڱ��ڣ�
�ٱ�һ��
�ڱ�����
��2���Ƚ������������Ľṹ���ҳ���ͬ��Ͳ�ͬ�㣮
��ͬ�㣺
��ͬ�㣺
��3����ȼ��þ���ŵ�������̼���ܾ���ȼ�գ���������þ�ͺ�ɫ��̼�����û�ѧʽ��ʾ�÷�Ӧ���̣�
��������1�����ʵķ�����кܶ��֣����������ʵ�״̬�����ʡ����ʡ���������Ԫ�صȷ��������ࣻ�����漰�ļ������ʣ��ɰ����ǵ����Ԫ�ء����Ƚ��з��࣮
��2�����ݽṹʾ��ͼ�����������
��3������þ�ڶ�����̼�е�ȼ��д����Ӧ�ķ���ʽ��
��2�����ݽṹʾ��ͼ�����������
��3������þ�ڶ�����̼�е�ȼ��д����Ӧ�ķ���ʽ��
����⣺��1����ˮ��H2O����������O2����������̼��CO2�������飨CH4�����������У�
�ٸ��������Ƿ�����Ԫ�ؿɷ�Ϊ�����࣬���鲻����Ԫ�أ�ˮ��������������̼��������Ԫ�أ�
�ڴ����ʷ���ĽǶȣ��������ڵ��ʣ�ˮ�����顢������̼�����ڻ����
��2�����������Ľṹʾ��ͼ��֪����ͬ���е��Ӳ�����ͬ�������ĵ������ȣ���ͬ���У����ڵ������������Եȣ�
��3����ȼ��þ���ŵ�������̼���ܾ���ȼ�գ���������þ�ͺ�ɫ��̼���÷�Ӧ�ķ���ʽ�ǣ�2Mg+CO2
2MgO+C��
�ʴ�Ϊ����1���ٶ�����Ԫ�أ�ˮ��������������̼���ڶ��ǻ����ˮ��������̼�����飮��2�������Ӳ�����ͬ��������������ͬ������������ͬ�����Բ�ͬ������3��2Mg+CO2
2MgO+C��
�ٸ��������Ƿ�����Ԫ�ؿɷ�Ϊ�����࣬���鲻����Ԫ�أ�ˮ��������������̼��������Ԫ�أ�
�ڴ����ʷ���ĽǶȣ��������ڵ��ʣ�ˮ�����顢������̼�����ڻ����
��2�����������Ľṹʾ��ͼ��֪����ͬ���е��Ӳ�����ͬ�������ĵ������ȣ���ͬ���У����ڵ������������Եȣ�
��3����ȼ��þ���ŵ�������̼���ܾ���ȼ�գ���������þ�ͺ�ɫ��̼���÷�Ӧ�ķ���ʽ�ǣ�2Mg+CO2
| ||
�ʴ�Ϊ����1���ٶ�����Ԫ�أ�ˮ��������������̼���ڶ��ǻ����ˮ��������̼�����飮��2�������Ӳ�����ͬ��������������ͬ������������ͬ�����Բ�ͬ������3��2Mg+CO2
| ||
������������һ�ֻ�ѧ�г��õ�ѧϰ������Ҫץס��������ݽ��з��࣬���ݲ�ͬ������Ľ��Ҳ��ͬ��Ҫѧ�ᰴ��ͬ�������ݶ����ʽ��з��࣮

��ϰ��ϵ�д�
�����Ŀ
��ͨ��ͭ����ͭ��п��ɣ��㷺���������ġ��ܲĵȣ�Ҳ���������е�����Ϊ�ⶨ��ͭ��ͭ������������ȡ��Ʒ10g�����Ĵ������м���ϡ����ʹ֮��ַ�Ӧ��ʵ�����ݼ�¼���±���
����������ݣ��ش��������⣺
��1����ͭ��ͭ����������Ϊ
��2��ǡ����ȫ��Ӧʱ����Һ�����ʵ����������ǣ�
| ��һ�� | �ڶ��� | ������ | ���Ĵ� | |
| ����ϡ���������/g | 10 | 10 | 10 | 10 |
| ʣ����������/g | 8.7 | 7.4 | 7 | 7 |
��1����ͭ��ͭ����������Ϊ
��2��ǡ����ȫ��Ӧʱ����Һ�����ʵ����������ǣ�
��Һ�����������ϢϢ��أ���Һ���������ճ�����ͻ�ѧʵ���еij����������±���������Һ�Ͱ�ˮ���ܶ��������ʵ������������ձ���20�棩��
����ϸ������ش��������⣺
��1��20��ʱ��������Һ�����ʵ���������������������Һ���ܶ��� �����������䣩����ˮ���ܶ��� ��������С�䣩
��2��ȡ12%��������Һ100g���Ƴ�6%����Һ����100g12%��������Һ�м�ˮ������
Ӧ 100g������ڡ�С�ڻ���ڣ���
��3����100g24%�İ�ˮ�м���100gˮ��ҡ�ȣ���Һ����� mL��������0.1����
| ��Һ�����ʵ���������/% | 4 | 12 | 16 | 24 | 28 |
| ������Һ���ܶ�/g/mL | 1.02 | 1.08 | 1.11 | 1.17 | 1.20 |
| ��ˮ���ܶ�/g/mL | 0.98 | 0.95 | 0.94 | 0.91 | 0.90 |
��1��20��ʱ��������Һ�����ʵ���������������������Һ���ܶ���
��2��ȡ12%��������Һ100g���Ƴ�6%����Һ����100g12%��������Һ�м�ˮ������
Ӧ
��3����100g24%�İ�ˮ�м���100gˮ��ҡ�ȣ���Һ�����