��Ŀ����
��ѧ������ѧϰ���о���ѧ����Ҫ���ߣ��밴Ҫ��������и��⣺
��1������Ԫ�ط��Żش�
������������ȱ �Ỽ���Ͳ��ͷ���������
�ڸ�����������Ҫ�ľ�������֮һ��Ϊ����ǿ����Ŀ��������������ʵ�ʩ�� �ʣ�
��2���������ӷ��Żش�
��ʳ�ε���Ҫ�ɷ����Ȼ��ƣ������� �������ӹ��ɵģ�
�ڼ����һЩ���ƵĻ�ѧ���ʣ�����Ϊ����Һ�ж����� ��
��3�����û�ѧʽ�ش�
��ũҵ���������������������� ��
�ڵ���Ǽ�ͥ����ҩ������е��ܼ��� ��
��4�����û�ѧ����ʽ�ش�
��������ˮ�������ԣ�ԭ���� ��
�������е�����Ʒ����ʴ���ܺã�ԭ���� ��
��1������Ԫ�ط��Żش�
������������ȱ
�ڸ�����������Ҫ�ľ�������֮һ��Ϊ����ǿ����Ŀ��������������ʵ�ʩ��
��2���������ӷ��Żش�
��ʳ�ε���Ҫ�ɷ����Ȼ��ƣ�������
�ڼ����һЩ���ƵĻ�ѧ���ʣ�����Ϊ����Һ�ж�����
��3�����û�ѧʽ�ش�
��ũҵ����������������������
�ڵ���Ǽ�ͥ����ҩ������е��ܼ���
��4�����û�ѧ����ʽ�ش�
��������ˮ�������ԣ�ԭ����
�������е�����Ʒ����ʴ���ܺã�ԭ����
���㣺Ԫ�صķ��ż�������,��ѧʽ����д������,��ѧ���ż�����Χ���ֵ�����,��д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ
ר�⣺��ѧ����������غ㶨��
��������1���Ӹ�Ԫ�ص��������ýǶȷ����ش𣻸��ݼط��ܴٽ�����������׳�����˴�Ӳ����ǿ�����溦�Ϳ���������������
��2������ʳ�εĹ��ɷ��������ݼ�ĸ��������
��3�����ݸ�����������һ���ü����������к������������ݵ���ǹ�̬��;ƾ�����Һ���з�����
��4�����ݶ�����̼����ˮ����̼����������������������ɺ����ý��з�����
��2������ʳ�εĹ��ɷ��������ݼ�ĸ��������
��3�����ݸ�����������һ���ü����������к������������ݵ���ǹ�̬��;ƾ�����Һ���з�����
��4�����ݶ�����̼����ˮ����̼����������������������ɺ����ý��з�����
����⣺��1������������ȱ�ƻỼ���Ͳ��ͷ������������ܴٽ�����������׳�����˴�Ӳ����ǿ�����溦�Ϳ��������������Ϊ����ǿ����Ŀ��������������ʵ�ʩ�üطʣ�
��2���Ȼ������������Ӻ������ӹ��ɵģ������һЩ���ƵĻ�ѧ���ʣ�����Ϊ����Һ�ж��������������ӣ�
��3��������������һ���ü����������кͣ���ʯ�ҵ���Ҫ�ɷ����������ƣ���һ�ּ��Ե����ʣ��������к������������������ṹ������ǹ�̬��;ƾ�����Һ�����оƾ����ܼ���
��4��������ˮ�������ԣ�ԭ���ǿ����еĶ�����̼����ˮ������̼�ᣮ��ѧ����ʽΪ��CO2+H2O�TH2CO3�������е�����Ʒ����ʴ���ܺã�ԭ����������������һ��������Ĥ��������Ʒ�������ã��仯ѧ����ʽΪ��4Al+3O2=2Al2O3��
�ʴ�Ϊ����1��Ca��K����2��Na+��OH-����3��Ca��OH��2��C2H5OH����4��CO2+H2O�TH2CO3��4Al+3O2=2Al2O3��
��2���Ȼ������������Ӻ������ӹ��ɵģ������һЩ���ƵĻ�ѧ���ʣ�����Ϊ����Һ�ж��������������ӣ�
��3��������������һ���ü����������кͣ���ʯ�ҵ���Ҫ�ɷ����������ƣ���һ�ּ��Ե����ʣ��������к������������������ṹ������ǹ�̬��;ƾ�����Һ�����оƾ����ܼ���
��4��������ˮ�������ԣ�ԭ���ǿ����еĶ�����̼����ˮ������̼�ᣮ��ѧ����ʽΪ��CO2+H2O�TH2CO3�������е�����Ʒ����ʴ���ܺã�ԭ����������������һ��������Ĥ��������Ʒ�������ã��仯ѧ����ʽΪ��4Al+3O2=2Al2O3��
�ʴ�Ϊ����1��Ca��K����2��Na+��OH-����3��Ca��OH��2��C2H5OH����4��CO2+H2O�TH2CO3��4Al+3O2=2Al2O3��
���������⿼��Ԫ�ص����ã����ʵ����ã����ʵĹ��ɣ���Һ�ijɷݣ����������ķ�����������Ʒ�ķ���ȣ�֪ʶ��࣬������ǿ��Ҫ�ι����գ�

��ϰ��ϵ�д�
�����Ŀ
����ҽѧԺ����ɽԺʿǿ����H7N9�����з���5����ʹ�ô�ƿ�����������֢�������л���Խ������Ч��Խ�ã�����ơ���Ҫ��Ч�ɷ���ç���ᣨC7H10O5�������й���ç����˵����ȷ���ǣ�������
| A������������ |
| B����7��̼Ԫ�ء�10����Ԫ�ء�5����Ԫ����� |
| C��174gç�����к���84g̼Ԫ�� |
| D��һ��ç��������к���5�������� |

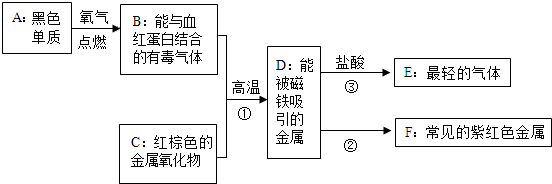
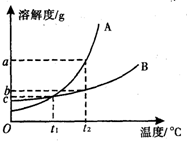 ��ͼ��A��B���ֹ������ʵ��ܽ�����ߣ�
��ͼ��A��B���ֹ������ʵ��ܽ�����ߣ� ̼�����������ᷴӦ�Ļ�ѧ����ʽΪ
̼�����������ᷴӦ�Ļ�ѧ����ʽΪ