��Ŀ����
����̼�������������ͬ��ע�Ļ��⣮
��1��Ϊ�������Ӿ������ЧӦ����ѧ�������о���ͼ��ʾ�Ķ�����̼��ѭ����ϵ����������ͼ�ش��������⣺
�ٽ����������ж�����̼����������������Ҫԭ����______��
�ڴ�ͼ��֪����һ�������£�ijЩ������л����______����ܡ����ܡ����ת����
��ˮ�ڹ�������������¿�ת��Ϊ������д����ѧ����ʽ______��������̼�������ڸ��ϴ��������������ɼ״���CH3OH����ˮ��д���÷�Ӧ�Ļ�ѧ����ʽ______��
�ܡ���̼����һ������������̬�ȣ������������ϡ���̼��Ҫ�����______ ������ţ���
A���������������� B���������������������������
C�������������?D������ʹ��β������װ�ã������к������ŷ�
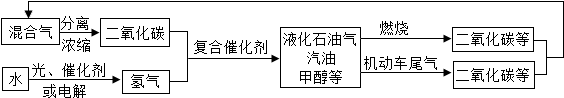
��2���Ƽ�С��ͬѧ�����һ�ֽ�CO2ת����ʵ��������ͼ��ʾ����ʵ��������CO2ת�������ղ���Ϊ______���ѧʽ����ͬ������һ�ֿ�ѭ�����õ�������______��
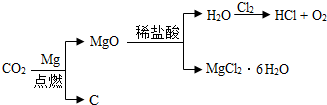
�⣺��1���ٺ�̼����ȼ��ȼ��ȼ��ʱ��������������̼�Ƕ�����̼�������Ҫԭ��
�ڴ�ͼʾ�п��Կ���������̼�������ڴ�����������ת���ɼ״�����������������л������ת����
��ˮ�ڹ�������������¿�ת��Ϊ��������ѧ����ʽΪ2H2O 2H2��+O2����������̼�������ڸ��ϴ��������������ɼ״���CH3OH����ˮ��д���÷�Ӧ�Ļ�ѧ����ʽCO2+3H2
2H2��+O2����������̼�������ڸ��ϴ��������������ɼ״���CH3OH����ˮ��д���÷�Ӧ�Ļ�ѧ����ʽCO2+3H2 CH3OH+H2O
CH3OH+H2O
�ܵ�̼������Ǽ��ٶ�����̼���ŷţ���Ҫ�ӽ��ܡ��ڵ�ͽ��������濼�ǣ�A���ⶨ��������������Ч�Ŀ��ƴ��������������̼������ֱ�ӹ�ϵ��B���ɼ�������ȼ�ϵ�ȼ�գ����ٶ�����̼�ŷţ����ϵ�̼���C�����Խ�Լ��Դ�ͱ�����Դ�����̼������ֱ�ӹ�ϵ��D��������Ч�ؽ��Ϳ�������Ⱦ�����̼������ֱ�ӹ�ϵ��
��2��ij��ѧ��ȤС���ͬѧ����˽�CO2ת����ʵ�飨ʵ��������ͼ��������ʵ��������CO2ת�������ղ���Ϊ C��O2��������뷴Ӧ����������HCl�����ڴ˹����п�ѭ�����õ�������HCl��
�ʴ�Ϊ����1���ٹ���ʹ�ÿ���ȼ�ϣ� ���ܣ� ��2H2O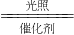 2H2��+O2����CO2+3H2
2H2��+O2����CO2+3H2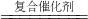 CH3OH+H2O�� ��B��
CH3OH+H2O�� ��B��
��2��C��O2��HCl��
��������1��������̼����������Ҫԭ���Ǻ�̼ȼ�ϵ�ȼ�գ��л��������֮�����ת�������ݷ�Ӧ���������д����ѧ����ʽʽ�����ݵ�̼������ٶ�����̼���ŷš��ӽ��ܡ��ڵ硢���������濼�ǣ�
��2����ʵ������ͼ����֪����ʵ��������CO2ת�������ղ���Ϊ C��O2��������뷴Ӧ����������HCl�����ڴ˹����п�ѭ�����õ�������HCl��
���������⿼��Ե�̼���õ����⣮��̼������һ���Ե��ܺĺ�Ч�ܵ�Ϊ��Ҫ�������Խ��ٵ����������ŷŻ�ýϴ�������¾��÷�չģʽ��
�ڴ�ͼʾ�п��Կ���������̼�������ڴ�����������ת���ɼ״�����������������л������ת����
��ˮ�ڹ�������������¿�ת��Ϊ��������ѧ����ʽΪ2H2O
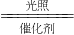 2H2��+O2����������̼�������ڸ��ϴ��������������ɼ״���CH3OH����ˮ��д���÷�Ӧ�Ļ�ѧ����ʽCO2+3H2
2H2��+O2����������̼�������ڸ��ϴ��������������ɼ״���CH3OH����ˮ��д���÷�Ӧ�Ļ�ѧ����ʽCO2+3H2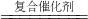 CH3OH+H2O
CH3OH+H2O�ܵ�̼������Ǽ��ٶ�����̼���ŷţ���Ҫ�ӽ��ܡ��ڵ�ͽ��������濼�ǣ�A���ⶨ��������������Ч�Ŀ��ƴ��������������̼������ֱ�ӹ�ϵ��B���ɼ�������ȼ�ϵ�ȼ�գ����ٶ�����̼�ŷţ����ϵ�̼���C�����Խ�Լ��Դ�ͱ�����Դ�����̼������ֱ�ӹ�ϵ��D��������Ч�ؽ��Ϳ�������Ⱦ�����̼������ֱ�ӹ�ϵ��
��2��ij��ѧ��ȤС���ͬѧ����˽�CO2ת����ʵ�飨ʵ��������ͼ��������ʵ��������CO2ת�������ղ���Ϊ C��O2��������뷴Ӧ����������HCl�����ڴ˹����п�ѭ�����õ�������HCl��
�ʴ�Ϊ����1���ٹ���ʹ�ÿ���ȼ�ϣ� ���ܣ� ��2H2O
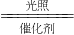 2H2��+O2����CO2+3H2
2H2��+O2����CO2+3H2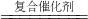 CH3OH+H2O�� ��B��
CH3OH+H2O�� ��B����2��C��O2��HCl��
��������1��������̼����������Ҫԭ���Ǻ�̼ȼ�ϵ�ȼ�գ��л��������֮�����ת�������ݷ�Ӧ���������д����ѧ����ʽʽ�����ݵ�̼������ٶ�����̼���ŷš��ӽ��ܡ��ڵ硢���������濼�ǣ�
��2����ʵ������ͼ����֪����ʵ��������CO2ת�������ղ���Ϊ C��O2��������뷴Ӧ����������HCl�����ڴ˹����п�ѭ�����õ�������HCl��
���������⿼��Ե�̼���õ����⣮��̼������һ���Ե��ܺĺ�Ч�ܵ�Ϊ��Ҫ�������Խ��ٵ����������ŷŻ�ýϴ�������¾��÷�չģʽ��

��ϰ��ϵ�д�
�����Ŀ








