��Ŀ����
����Ŀ���˵����Ϊ 1~18 ��Ԫ�ص�ԭ�ӽṹʾ��ͼ����Ϣ���£���ش��������⡣

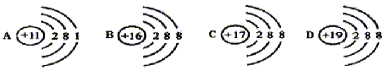
��1��ԭ������Ϊ 11 ��Ԫ������_______����������ǽ�������Ԫ�أ�λ�ڵ� 2 ���ڵ�VA ���Ԫ��������________��
��2����Ԫ�غ�̼Ԫ�������ƵĻ�ѧ���ʣ�����Ϊ��ԭ�Ӻ�̼ԭ�ӵ�__________��ͬ��
��3��һ���˵�����Ӳ�����ͬ��ԭ�ӣ�ϡ��������⣩��ԭ�Ӱ뾶����ԭ�������ĵ�������С��������������ͬ��ԭ�ӣ�ԭ�Ӱ뾶����Ӳ��������Ӷ�������Ƚϣ���ԭ�ӡ���ԭ�Ӻ���ԭ�ӵİ뾶�ɴ�С��˳��Ϊ____________��
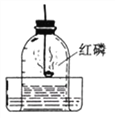
��4��ԭ������Ϊ 15 ��Ԫ����ɵĵ��ʳ����������ⶨ�����������ĺ�����ʵ�飨��ͼ��ʾ����ʵ���иõ��ʷ�����Ӧ�Ļ�ѧ����ʽΪ__________�����ڸ�ʵ�飬����˵����ȷ����________��

A����ʵ���˵��������������ˮ
B��ȼ�ճ��еĹ�����Ի���ľ̿
C��ȼ�ճ��еĹ���������ᵼ�²ⶨ���ƫС
D��ʵ������е��ɼ�δ�н��ᵼ�²ⶨ���ƫС
��5��A ����Ϊ���˻�泣��ȼ�ϣ������� A �� B �����г��ȼ�գ�������Ӧ��A+5B ![]() 3C+4D��������ʾ��ͼ������ʾ����
3C+4D��������ʾ��ͼ������ʾ����

����������Ӧ���ṩ�������������Ե�����Ϊ________���ѧʽ����
��A �����и�Ԫ�ص�������Ϊ___________��
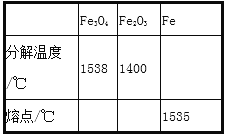
���𰸡������� �� ���������� ��ԭ�ӣ���ԭ�ӣ���ԭ�� 4P+5O2 ![]() 2P2O5�� AC O2�� ̼Ԫ�ء���Ԫ�ص�������Ϊ 9:2
2P2O5�� AC O2�� ̼Ԫ�ء���Ԫ�ص�������Ϊ 9:2
��������
��1��ԭ������Ϊ 11 ��Ԫ������Ԫ�أ����ڽ���Ԫ�أ����������
λ�ڵ� 2 ���ڵ�VA ���Ԫ�������ǵ��������
��2��Ԫ�ؾ������ƵĻ�ѧ��������ΪԪ��ԭ�ӵ�������������ͬ������������������
��3�����Ӳ�����ͬ��ԭ�ӣ�ϡ��������⣩��ԭ�Ӱ뾶����ԭ�������ĵ�������С������ͬһ���ڵ���Ԫ�ص���ԭ�Ӱ뾶����ͬһ���ڵ���Ԫ�ص���ԭ�ӵİ뾶��������������ͬ��ԭ�ӣ�ԭ�Ӱ뾶����Ӳ��������Ӷ�������������������ͬ����ԭ�ӵİ뾶������ԭ�ӵİ뾶����������ԭ�ӵİ뾶��С��������ԭ�ӣ���ԭ�ӣ���ԭ�ӣ�������ԭ�ӣ���ԭ�ӣ���ԭ����
��4��ԭ������Ϊ 15 ��Ԫ����ɵĵ��������ʡ��������ڵ�ȼ�������·�Ӧ�������������ס��ʻ�ѧ����ʽдΪ��4P+5O2 ![]() 2P2O5��
2P2O5��
A����ʵ���������ƿ�ڵ�ˮֻռƿ�ݻ���1/5����˵��������������ˮ��ѡ����ȷ��
B��ȼ�ճ��еĹ��岻���Ի���ľ̿����Ϊľ̼������ȼ�պ����ɶ�����̼������ʵ�����������ţ�ѡ�����C��ȼ�ճ��еĹ���������ᵼ�������ƿ������ʣ�࣬ʹ�ⶨ���ƫС��ѡ����ȷ��D��ʵ������е��ɼ�δ�н��ᵼ��ƿ�����͵������ų�ƿ�⣬ʹ�ⶨ���ƫ��ѡ�����ѡAC��
��5���������غ㶨�ɿ�֪��������Ӧ��AΪ���飬BΪ������CΪ������̼��DΪˮ�����о��������Ե���������������ѧʽΪO2������O2��
AΪ���飬��ѧʽΪC3H8������̼Ԫ�ء���Ԫ�ص�������Ϊ��12��3������1��8��= 9��2������9��2��

 ��������ܸ�ϰϵ�д�
��������ܸ�ϰϵ�д�����Ŀ����֪п��������Է�����Ӧ��������п��������ij�о���ѧϰС����̽����Ӱ��п�����ᷴӦ���������ء�ʱ������һ���������������л�������ͭ��Һʱ���������������ʸ��졣
���������ϣ���п�����ᷴӦ�����ű���ʽZn + H2SO4 = ZnSO4 + H2
��п������ͭ��Һ��Ӧ�����ű���ʽZn + CuSO4 = ZnSO4 + Cu
��������Һ�к���H+����������ӣ�����ѧ������ʾ_____________��������ͭ��Һ�к���Cu2+����������ӣ�����п��Һ�к���Zn2+����������ӡ�
��������⣩��ʲô���ӵ��²������������ʼӿ죿
��������裩��ͬѧ��������ͭ�����йأ�
��ͬѧ������������������йأ�
��ͬѧ��Ϊ�ҵIJ��벻��ѧ��������_________��
��ʵ����֤��д�������ٵ�����__________����A��B������������ʵ�飺
����һ�����װ�������ԡ�����AB����Ĵָ��ס���ܿڣ��ӳ���©����ˮ��һ���߶ȣ����ã��۲쵽_______����Ϊ���������á�
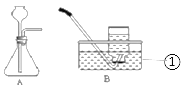
���������ȡ2����������Ϊ20%��ϡ�����20mL����һ�ݼ�����������Ϊ10%������ͭ��Һ1mL���ڶ��ݼ�����������Ϊ10%������п��Һ1mL����ȡ2����״��ͬп����1g���ֱ���������Һ��Ӧ����Ӧװ����ͼA��ʾ���ô�װ��ʵ��ʱ������©���¶�Ҫ����Һ�����£�Ŀ����__________��
����������B�ռ����壬�ֱ��¼�ռ�һƿ���壨����ƿ����Ϊ125mL�������ʱ�䣨���±�����ʲôʱ�俪ʼ�ռ����壿__________����������������ȡ��������ݡ���
��� | 20%��ϡ�����20mL | п�� | �ռ�1ƿ���������ʱ�� |
�� | ������������Ϊ10%������ͭ��Һ1mL | 1g | 2��10�� |
�� | ������������Ϊ10%������п��Һ1mL | 1g | 3��01�� |
���ó����ۣ������л�������ͭ��Һʱ���������������ʼӿ���__________�йء�ʵ����м�����������Ϊ10%������п��Һ1mL��������__________��
����˼���ɣ���ͬѧ��Ϊ��п���������ͭ��������Һ��Ӧʱ���������������ʽϿ������ͭ�����йأ������ɵ�������__________��
������̽�����������һ��ʵ�鷽����֤��ͬѧ�Ĺ۵��Ƿ���ȷ��
��� | ʵ�鲽�� | ʵ������Ƚ� | ʵ����� |
�� | ȡ1gп������ƿ�У� ����20mL 20%��ϡ���� | __________ | �������������ʽϿ���ͭ�����й� |
�� | __________ |
����Ŀ���ּס�������ѧС�鰲װ����������ͬװ�ã�ͨ���ⶨ������ͬ�����������ʱ�䳤����̽��Ӱ��H2O2�ֽ����ʵ����ء�

��1�����з����ܼ���װ�������Ե���_______��
A����ע���������̶������Һ©����ע����������ˮ����Һ©�������������Һ©���������γ��ȶ���ˮ���������������ã���֮�����Բ
B���رշ�Һ©��������������ƿ�����ע�����������ƣ����ƣ���ֹͣ���Ⱥ�ע���������ص�ԭ��λ�ã������������ã���֮�����Բ
C���رշ�Һ©��������������ע�����������ƣ����ƣ���ֹͣ������ע�����������ƶ��������������ã���֮�����Բ
��2����С��������ʵ����Ʒ���������±���
ʵ���� | ʵ��Ŀ�� | �¶� | ���� | Ũ�� |
����ʵ��� | ̽��_________��H2O2�ֽ����ʵ�Ӱ�� | 25�� | �������� | 10mL 2%H2O2 |
����ʵ��� | 25�� | �������� | 10mL 5%H2O2 |
��3���ס�����С��ó�����ͼ���ݡ�

�ټ���ʵ��ó������ݿ�֪��Ũ��Խ��H2O2�ֽ�����_________;
���������о����ᡢ���H2O2�ֽ�Ӱ�����ص� ������ͬ������, H2O2��_______����ᡱ����Ի����·ų��������ʽϿ�