��Ŀ����
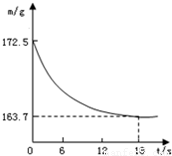
 ij��˾�������Ĵ����Ʒ�о����ֻ�����Ȼ������ʣ�Ϊ�ⶨ��Ʒ��̼���Ƶ�����������20��ʱ����ȡ�ò�Ʒ��Ʒ26.5g�����뵽ʢ��һ������ϡ������ձ��У�̼������ϡ����ǡ����ȫ��Ӧ��������ȫ�ݳ����õ�������NaCl��Һ����Ӧ�����þ�����������ձ��ڻ�����������m���뷴Ӧʱ�䣨t����ϵ��ͼ��ʾ��
ij��˾�������Ĵ����Ʒ�о����ֻ�����Ȼ������ʣ�Ϊ�ⶨ��Ʒ��̼���Ƶ�����������20��ʱ����ȡ�ò�Ʒ��Ʒ26.5g�����뵽ʢ��һ������ϡ������ձ��У�̼������ϡ����ǡ����ȫ��Ӧ��������ȫ�ݳ����õ�������NaCl��Һ����Ӧ�����þ�����������ձ��ڻ�����������m���뷴Ӧʱ�䣨t����ϵ��ͼ��ʾ����
��1������CO2��������
��2���ô�����Ʒ��Na2CO3������������
��������1�����������غ㶨�ɼ�ͼʾ����������CO2��������
��2�����ݷ�Ӧ����ʽ������CO2�����������㴿����Ʒ��Na2CO3����������������ô�����Ʒ��Na2CO3������������
��2�����ݷ�Ӧ����ʽ������CO2�����������㴿����Ʒ��Na2CO3����������������ô�����Ʒ��Na2CO3������������
����⣺��1�����ɶ�����̼������Ϊ172.5g-163.7 g=8.8 g��
��2���裺��Ʒ��̼���Ƶ�����Ϊx
Na2CO3+2HCl�T2NaCl+CO2��+H2O
106 44
x 8.8g
=
��� x=21.2 g
��ô�����Ʒ��̼���Ƶ���������Ϊ
��100%=80%��
�𣺣�1�����ɶ�����̼������8.8 g��
��2���ô�����Ʒ��Na2CO3��������Ϊ80%��
��2���裺��Ʒ��̼���Ƶ�����Ϊx
Na2CO3+2HCl�T2NaCl+CO2��+H2O
106 44
x 8.8g
| 106 |
| x |
| 44 |
| 8.8g |
��ô�����Ʒ��̼���Ƶ���������Ϊ
| 21.2g |
| 26.5g |
�𣺣�1�����ɶ�����̼������8.8 g��
��2���ô�����Ʒ��Na2CO3��������Ϊ80%��
��������������ͼ������Ӧ����ʽ�������ͽ����ѧ�����е��й����⣬Ҫ��ѧ���н�ǿ��ʶͼ���������ݷ���������

��ϰ��ϵ�д�
�����Ŀ
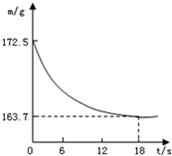
��9�֣�ij��˾�������Ĵ����Ʒ�о����ֻ�����Ȼ������ʡ�Ϊ�ⶨ��Ʒ��̼���Ƶ�����������20��ʱ����ȡ�ò�Ʒ��Ʒ26.5g�����뵽ʢ��һ������ϡ������ձ��У�̼������ϡ����ǡ����ȫ��Ӧ��������ȫ�ݳ����õ�������NaCl��Һ����Ӧ�����þ�����������ձ��ڻ�����������m���뷴Ӧʱ�䣨t����ϵ����ͼ��ʾ��
��1������CO2��������
��2���ô�����Ʒ��Na2CO3������������
��3���������������������������е� ��ֻ����ĸ��ţ������㣬�ɶ�ѡ����

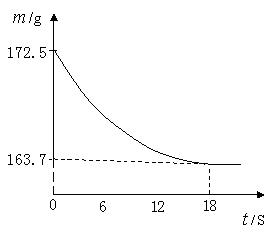
��1������CO2��������
��2���ô�����Ʒ��Na2CO3������������
��3���������������������������е� ��ֻ����ĸ��ţ������㣬�ɶ�ѡ����
| A���ô�����Ʒ��NaCl������������ |
| B����Ӧ����ϡ������������������� |
| C����Ӧ��������Һ��NaCl������������ |
| D����Ӧ����ˮ�������� |
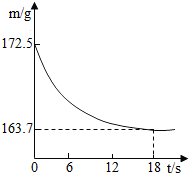 ij��˾�������Ĵ����Ʒ�о����ֻ�����Ȼ������ʣ�Ϊ�ⶨ��Ʒ��̼���Ƶ�����������20��ʱ����ȡ�ò�Ʒ��Ʒ26.5g�����뵽ʢ��һ������ϡ������ձ��У�̼������ϡ����ǡ����ȫ��Ӧ��������ȫ�ݳ����õ�������NaCl��Һ����Ӧ�����þ�����������ձ��ڻ�����������m���뷴Ӧʱ�䣨t����ϵ����ͼ��ʾ��
ij��˾�������Ĵ����Ʒ�о����ֻ�����Ȼ������ʣ�Ϊ�ⶨ��Ʒ��̼���Ƶ�����������20��ʱ����ȡ�ò�Ʒ��Ʒ26.5g�����뵽ʢ��һ������ϡ������ձ��У�̼������ϡ����ǡ����ȫ��Ӧ��������ȫ�ݳ����õ�������NaCl��Һ����Ӧ�����þ�����������ձ��ڻ�����������m���뷴Ӧʱ�䣨t����ϵ����ͼ��ʾ�� ij��˾�������Ĵ����Ʒ�о����ֻ�����Ȼ������ʣ�Ϊ�ⶨ��Ʒ��̼���Ƶ�����������20��ʱ����ȡ�ò�Ʒ��Ʒ26.5g�����뵽ʢ��һ������ϡ������ձ��У�̼������ϡ����ǡ����ȫ��Ӧ��������ȫ�ݳ����õ�������NaCl��Һ����Ӧ�����þ�����������ձ��ڻ�����������m���뷴Ӧʱ�䣨t����ϵ��ͼ��ʾ��
ij��˾�������Ĵ����Ʒ�о����ֻ�����Ȼ������ʣ�Ϊ�ⶨ��Ʒ��̼���Ƶ�����������20��ʱ����ȡ�ò�Ʒ��Ʒ26.5g�����뵽ʢ��һ������ϡ������ձ��У�̼������ϡ����ǡ����ȫ��Ӧ��������ȫ�ݳ����õ�������NaCl��Һ����Ӧ�����þ�����������ձ��ڻ�����������m���뷴Ӧʱ�䣨t����ϵ��ͼ��ʾ��