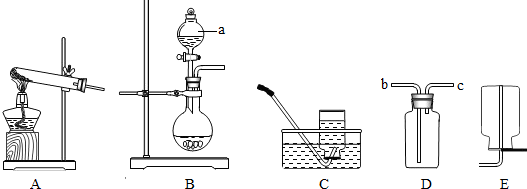
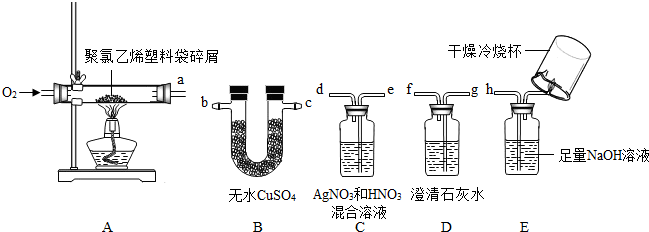
��Ŀ����
 ��1����ˮ��H2O����˫��ˮ��H2O2������ˮ��D2O����D����ʾԭ�Ӻ�����һ�����Ӻ�һ�����ӵ���ԭ�ӣ��������ʣ���������ɵĽǶȿ�������������������Ԫ�ع���
��1����ˮ��H2O����˫��ˮ��H2O2������ˮ��D2O����D����ʾԭ�Ӻ�����һ�����Ӻ�һ�����ӵ���ԭ�ӣ��������ʣ���������ɵĽǶȿ�������������������Ԫ�ع���2
2
�֣��ӻ�ѧ���ʵĽǶȿ���H2O2
H2O2
��ѡ�H2O2����D2O������ˮ�Ļ�ѧ�����нϴ�IJ��죮��2�����С�ˮ���У�һ�����ڴ��������
��
��
������ˮ ����ˮ �۱�ˮ����� ����ˮ �ݿ�Ȫˮ ����ˮ
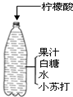
��3��С������ͼ�䷽����һƿ�����������ˮ������ͼ����Ϣ�ش�
�ٰ��ǵ���Ҫ�ɷ������ǣ��仯ѧʽΪ
C12H22O11
C12H22O11
��С�մ��к��еĽ���Ԫ�ص���������Ԫ��
��Ԫ��
���ڴ���ˮƿ�Dz����������Ҫ�ɷ���
CO2
CO2
������������������С�մ�
С�մ�
��Ӧ���ɣ���4��Ҫ�ⶨij�زɼ�������ˮ�Ƿ�Ϊ���꣬�����ṩ���Լ�������������ȡ��
C
C
��A����ɫʯ����Һ B����ɫ��̪��Һ C��pH�ƣ�
��������1�����ݻ�ѧʽ���з�����Ԫ����ɣ����ʵĽṹ�������ʵ����ʣ�
��2�����ݡ�ˮ����ɣ��������ʵ����
��3����д�����ǻ�ѧʽ������С�մ�Ļ�ѧʽ������
�ڸ���С�մ�������������ʷ�����
��4������������pHС��5.6�Ľ�ˮ��֪Ҫ��֤���Ƿ���������Ҫ�ⶨ��Һ��pH���ݴ˷������ɣ�
��2�����ݡ�ˮ����ɣ��������ʵ����
��3����д�����ǻ�ѧʽ������С�մ�Ļ�ѧʽ������
�ڸ���С�մ�������������ʷ�����
��4������������pHС��5.6�Ľ�ˮ��֪Ҫ��֤���Ƿ���������Ҫ�ⶨ��Һ��pH���ݴ˷������ɣ�
����⣺��1����ˮ��H2O����˫��ˮ��H2O2������ˮ��D2O���Ļ�ѧʽ��֪�����⡢������Ԫ����ɣ����ʵĽṹ�������ʵ����ʣ��ӻ�ѧ���ʵĽǶȿ���˫��ˮ ��ˮ�Ļ�ѧ�����нϴ�IJ��죻
��2������ˮ����ˮ����ˮ����Ȫˮ������ˮ�ж������������ʣ����ڻ�����ˮ���������һ��������ɵģ����ڴ����
��3���ٰ��ǵ���Ҫ�ɷ������ǣ��仯ѧʽΪC12H22O11��С�մ��к��еĽ���Ԫ�ص���������Ԫ�أ�
�ڴ���ˮƿ�Dz����������Ҫ�ɷ���CO2������������������С�մ�Ӧ���ɣ�
��4��pH���ܽ�ȷ�ز�����Һ�����ǿ���ij̶ȣ����Լ���ij���Ƿ������������pH�ƽ��в�����
�ʴ�Ϊ����1��2��H2O2����2���ۣ���3����C12H22O11����Ԫ�أ���CO2��С�մ�4��C��
��2������ˮ����ˮ����ˮ����Ȫˮ������ˮ�ж������������ʣ����ڻ�����ˮ���������һ��������ɵģ����ڴ����
��3���ٰ��ǵ���Ҫ�ɷ������ǣ��仯ѧʽΪC12H22O11��С�մ��к��еĽ���Ԫ�ص���������Ԫ�أ�
�ڴ���ˮƿ�Dz����������Ҫ�ɷ���CO2������������������С�մ�Ӧ���ɣ�
��4��pH���ܽ�ȷ�ز�����Һ�����ǿ���ij̶ȣ����Լ���ij���Ƿ������������pH�ƽ��в�����
�ʴ�Ϊ����1��2��H2O2����2���ۣ���3����C12H22O11����Ԫ�أ���CO2��С�մ�4��C��
������������Ҫ����ѧ���Ի�ѧ�������д��������������Ŀ��ƼȰ����Ի�ѧ����������˽⣬�ֿ�����ѧ���Ի�ѧ���ŵ���д������ȫ�棬ע�ػ�����

��ϰ��ϵ�д�
 ��ս�п�����ϵ�д�
��ս�п�����ϵ�д�
�����Ŀ