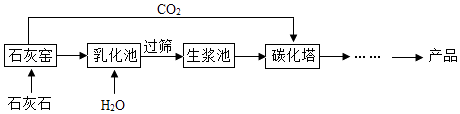
��Ŀ����
����Ŀ��������ʵ������ȡ����ij����������ش��������⣮ 
��1�����Ϊc������������ ��
��2��ʵ�����ø��������ȡO2 �� ������ˮ���ռ��� ��д����Ӧ�ķ���ʽ��
����װ��ȡװ��ʱ����ѡ����ͼ�е�b��d��e��g��m�� ��
��3����CaCO3+2HCl�TCaCl2+H2O+CO2����Ӧ��ȡCO2 �� ���������ſ������ռ��� �����ô���Ϊ90%�Ĵ���ʯ100g������ϡ���ᷴӦ�����Ƶ�CO2g��
�ڼ���CO2�Ƿ��ռ����ķ����� ��
���𰸡�
��1������©��
��2��2KMnO4 ![]() K2MnO4+MnO2+O2����f��i
K2MnO4+MnO2+O2����f��i
��3��39.6����ȼ�ŵ�ľ������ƿ�ڣ����۲쵽ľ��Ϩ��˵���ռ�����
���������⣺��1�����Ϊc�����������dz���©�����������©������2����������ڼ��ȵ������·ֽ�Ϊ����ء��������̺��������ü��ȸ��������ȡO2 �� ������ˮ���ռ�ʱ��Ҫ���������Թܡ�����̨�������ܵĵ�����Ƥ�����ƾ��ơ�ˮ�ۺͼ���ƿ�������2KMnO4 ![]() K2MnO4+MnO2+O2������f��i����3���ٽ⣺�����ɶ�����̼������Ϊx��
K2MnO4+MnO2+O2������f��i����3���ٽ⣺�����ɶ�����̼������Ϊx��
CaCO3+ | 2HCl�T | CaCl2+H2O+ | CO2�� |
100 | 44 | ||
100g��90% | x |
![]()
x=39.6g
���39.6�����ռ�һƿ������̼����ȼ�ŵ�ľ������ƿ�ڣ����۲쵽ľ��Ϩ��˵���ռ����ˣ������ȼ�ŵ�ľ������ƿ�ڣ����۲쵽ľ��Ϩ��˵���ռ����ˣ�
�����㾫����ͨ�����������������ȡװ�ú��������ռ����������շ���װ�ã��̹̼����͡���Һ�������ͣ��ռ�װ�ã��������ʵ��ܶȡ��ܽ��ԣ���ˮ���ռ�ʱ�������ݾ�������ð��ʱ���ռ����տ�ʼ�ų������Թ��еĿ����������ſ������ռ�����ʱ�������쵽����ƿ�ײ������Խ����⣮

 �����ҵ��ٿ���������������ϵ�д�
�����ҵ��ٿ���������������ϵ�д�