��Ŀ����
 ijѧУ��ѧ��ȤС��ͬѧ��ѧϰ����ȡ������һ�����ݺ�����ʵ��������5%H2O2��Һ������O2���ô����ǵ�ľ�����飬ľ�����Ѹ�ȼ��ͬѧ�Ǿ�������۲죬���������ԭ����
ijѧУ��ѧ��ȤС��ͬѧ��ѧϰ����ȡ������һ�����ݺ�����ʵ��������5%H2O2��Һ������O2���ô����ǵ�ľ�����飬ľ�����Ѹ�ȼ��ͬѧ�Ǿ�������۲죬���������ԭ�������ɵĴ���ˮ������ʵ����ɵ��˸���
���ɵĴ���ˮ������ʵ����ɵ��˸���
��Ϊ����֤�˲����Ƿ���ȷ����ȤС���ͬѧ�����������ۣ��ƶ���ʵ�鷽����������ʵ�飮��һ��ͬѧ��ȡ����H2O2��ҺŨ�ȵķ�������10%��H2O2��Һ���ȣ����һ��ʱ����ô����ǵ�ľ�����飬ľ����ȼ��
�ڶ����ͬѧ��5%��H2O2��Һ�м���2����������������Һ������һ��ʱ����ô�����ľ�����飬ľ����ȼ��
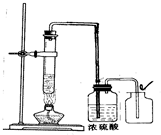
������ͬѧ��ʵ��װ���Ͻ����˸Ľ���Ч���ܺã���ͼ����
��ʵ������ϣ�ͬѧ�Ǿ����������ۣ��ó���һЩ����
���ɵ�һ��ͬѧ��ʵ��ɵó���Ӱ��H2O2�ֽ����O2�����Ķ��ټ��ֽ����������֮һ��
H2O2��ҺŨ�ȵĴ�С
H2O2��ҺŨ�ȵĴ�С
�����ɵڶ���ͬѧ��ʵ��ɷ����ó�����������������ÿ�����
�ٽ�H2O2�ֽ⣬����ˮ������
�ٽ�H2O2�ֽ⣬����ˮ������
���۵�����ͬѧ��ʵ����֤��ʵ��ǰͬѧ�Dz������ȷ�ԣ�����Ũ�����������
Ũ���������������е�ˮ������ʹ������Ũ������
Ũ���������������е�ˮ������ʹ������Ũ������
��������ľ������ȼ����ԭ����ȼ����������û��ͬʱ�߱�������������жϵ���ľ�����ܸ�ȼ����Ҫ�����������л��е�ˮ�йأ��Ա�ʵ��Ľ���ʵ���з����˸ı�����أ����������صĸı�Բ������������ٶȵ�Ӱ�죬���ɳ������ĸı�������������ӵ�ԭ��
����⣺�����ʵ�鶼���ڽ�������������⣬ʵ��������ȷ���жϷ�Ӧ�õ��������л��е�ˮ���������ǵ���ľ�����ܸ�ȼ��ԭ����ˣ��ò��뼴Ϊ������ˮ������ʵ������˸��ţ�
��1��ʵ�����10%��H2O2��Һ���ȣ��۲쵽ľ����ȼ��˵��Ӱ��H2O2�ֽ�������������Ķ��ټ��ֽ����������ΪH2O2��ҺŨ�ȵĴ�С��
��2��ʵ�������5%��H2O2��Һ�м���2����������������Һ���۲쵽ľ����ȼ�����ƶ��������������ƶ������̵Ĵ����û�������ˮ��������Ŀ�ģ�
��3��ʵ��Ѳ���������ͨ��������ˮ�Ե�Ũ���ᣬ�õ���������壬�۲쵽ľ����ȼ����˵��Ũ���������������е�ˮ������ʹ������Ũ������
�ʴ�Ϊ��
����ˮ������ʵ������˸��ţ�
��1��H2O2��ҺŨ�ȵĴ�С��
��2���ٽ�H2O2�ֽ⣬����ˮ��������
��3��Ũ���������������е�ˮ������ʹ������Ũ������
��1��ʵ�����10%��H2O2��Һ���ȣ��۲쵽ľ����ȼ��˵��Ӱ��H2O2�ֽ�������������Ķ��ټ��ֽ����������ΪH2O2��ҺŨ�ȵĴ�С��
��2��ʵ�������5%��H2O2��Һ�м���2����������������Һ���۲쵽ľ����ȼ�����ƶ��������������ƶ������̵Ĵ����û�������ˮ��������Ŀ�ģ�
��3��ʵ��Ѳ���������ͨ��������ˮ�Ե�Ũ���ᣬ�õ���������壬�۲쵽ľ����ȼ����˵��Ũ���������������е�ˮ������ʹ������Ũ������
�ʴ�Ϊ��
����ˮ������ʵ������˸��ţ�
��1��H2O2��ҺŨ�ȵĴ�С��
��2���ٽ�H2O2�ֽ⣬����ˮ��������
��3��Ũ���������������е�ˮ������ʹ������Ũ������
���������ڽ�����Ľ���ʵ��������Ľ������Ҫ��ʵ�������еĸı�����ȷ�İ��գ�������ʵ����������������������

��ϰ��ϵ�д�
 �Ƹ������������ϵ�д�
�Ƹ������������ϵ�д�
�����Ŀ