��Ŀ����
������һ���ᣬ���ᾧ�壨H2C2O4•2H2O��������ˮ���۵�ϵͣ����Ȼ��ۻ��������ͷֽ⡣���ᣨH2C2O4�����������Ƶķ�Ӧ��H2C2O4+Ca(OH)2=CaC2O4��(��ɫ)+2H2O��
���������ۣ�
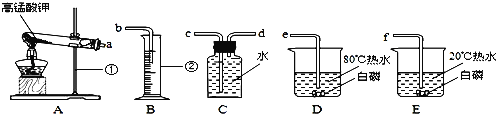
ʵ���ҿ��ü��Ȳ��ᾧ��ֽ�ķ������CO

��1���ȼ��Ȳ��ᾧ������CO��CO2��H2O���仯ѧ����ʽ��_____��
��2���������ͼװ���ռ�CO������Ӧ��_____�˽��루ѡ�a����b������
��ʵ�鷴˼��
��3������Ϊ��ͼ���Թܿ�Ӧ��������б����ʦ��ͬѧ���ۺ�һ����Ϊװ������ȷ�ģ�������_____��
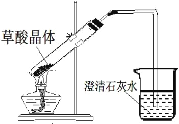
��4��ʵ���й۲쵽����ʯ��ˮ����ǣ�����Ϊһ�����ɲ��ᾧ�����ȷֽ������CO2�����£�����Ϊ�ҵĽ��۲����ܣ�������_____��
���������ӣ�����Ӫ���ḻ�������˶���ͬʳ���ý�ʯ����Ҫ�ɷֲ���ƾ��壩��С��ͬѧ�Բ���ƾ�������ʼ���ɲ�������Ȥ��
������̽��������ͼװ�ý�����ƾ��壨CaC2O4��xH2O����Ʒ���¼��ȣ�ʹ����ȫ�ֽⲢ������������塣
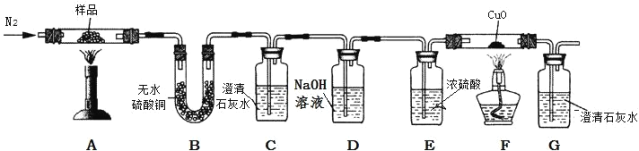
���������ۣ�
��5��B�й۲쵽_____����˵����Ӧ������ˮ��
��6��C��G�г���ʯ��ˮ������ǣ�˵����Ӧ������_____��_____���塣
��7����ͬѧ��Ϊ�������۲��Ͻ��������ܵó���һ����̼���ɵĽ��ۡ���˵������_____��
�����ȷ����
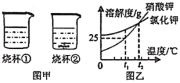
��8�������ȷ����ǶԲ���ƾ��壨CaC2O4��xH2O�������ȷֽ⣬���������ݣ����Ƴɹ����������ֽ��¶ȵĹ�ϵ��ͼ��
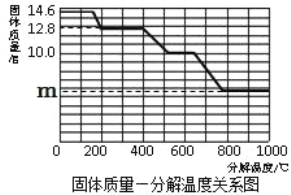
���¶�Ϊ200������ʱ������ȫ��ʧȥ�ᾧˮ�������нᾧˮ������Ϊ_____g��
�ڼ���CaC2O4��xH2O�е�x��CaC2O4����Է���������128����x=_____��
��800������ʱ���������������Ϊ�����ͼ��m��ֵ��_____��
��д������������12.8g��Ϊ10.0gʱ�Ļ�ѧ����ʽ_____��
 ����ʦ��Сһ����ʦ������ҵϵ�д�
����ʦ��Сһ����ʦ������ҵϵ�д�