��Ŀ����
���û�ѧ����ʽ������������
��1��Сӱͬѧ��̽��������̼����ʱ����������̼����ͨ����ɫʯ����Һ��ʯ����Һ�ܿ���ɫ��ԭ����
��2�����ڴ��ʯ��ˮ�ĮJ������һ�㱡Ĥ���ñ�Ĥ�γɵ�ԭ����
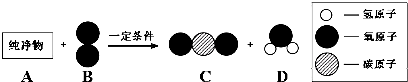
��3��������A����������ȼ�ϣ���һ��������A��B����ͼ��ʾ��ַ�Ӧ������C��D����֪A��һ�������к���3��̼ԭ�ӣ���A����Է�������Ϊ44����÷�Ӧ�Ļ�ѧ����ʽΪ

��1��Сӱͬѧ��̽��������̼����ʱ����������̼����ͨ����ɫʯ����Һ��ʯ����Һ�ܿ���ɫ��ԭ����
H2O+CO2�TH2CO3
H2O+CO2�TH2CO3
�������������⣬��ҵ�ϳ���ϡ������������Fe2O3+6HCl�T2FeCl3+3H2O
Fe2O3+6HCl�T2FeCl3+3H2O
����2�����ڴ��ʯ��ˮ�ĮJ������һ�㱡Ĥ���ñ�Ĥ�γɵ�ԭ����
CO2+Ca��OH��2�TCaCO3��+H2O
CO2+Ca��OH��2�TCaCO3��+H2O
����ʵ�����ﳣ��ϡ������ϴ��������Ӧ�Ļ�ѧ����ʽΪCaCO3+2HCl�TCaCl2+H2O+CO2��
CaCO3+2HCl�TCaCl2+H2O+CO2��
����3��������A����������ȼ�ϣ���һ��������A��B����ͼ��ʾ��ַ�Ӧ������C��D����֪A��һ�������к���3��̼ԭ�ӣ���A����Է�������Ϊ44����÷�Ӧ�Ļ�ѧ����ʽΪ
C3H8+5O2
3CO2+4H2O
| ||
C3H8+5O2
3CO2+4H2O
��
| ||
���������ȸ��ݷ�Ӧԭ���ҳ���Ӧ��������Ӧ���������ݻ�ѧ����ʽ����д���������������д���ɣ�
����⣺��1��������̼��ˮ��Ӧ����̼�ᣬ̼����ʹ��ɫʯ����Һ���ɫ����Ӧ�Ļ�ѧ����ʽΪ��H2O+CO2�TH2CO3���������Ҫ�ɷ����������������ᷴӦ�����Ȼ�����ˮ����Ӧ�Ļ�ѧ����ʽ�ǣ�Fe2O3+6HCl�T2FeCl3+3H2O��
��2��ʯ��ˮ�е��������������ƣ���������еĶ�����̼��Ӧ����̼��ƺ�ˮ��̼�����һ�ֲ�����ˮ�İ�ɫ���ʣ���ƿ�г��γ�һ���Ĥ����Ӧ�Ļ�ѧ����ʽΪ��CO2+Ca��OH��2�TCaCO3��+H2O��̼�������ϡ���ᷴӦ�����Ȼ��ơ�ˮ�Ͷ�����̼����Ӧ�Ļ�ѧ����ʽΪ��CaCO3+2HCl�TCaCl2+H2O+CO2����
��3����֪A��һ�������к���3��̼ԭ�ӣ���A����Է�������Ϊ44�����������غ㶨�ɣ�A��һ�������к���8����ԭ�ӣ�B���ӵĻ�ѧʽΪ��O2��C�Ļ�ѧʽΪ��CO2��D�Ļ�ѧʽΪ��H2O����Ӧ�Ļ�ѧ����ʽΪ��Fe2O3+6HCl=2FeCl3+3H2O��
�ʴ�Ϊ����1��H2O+CO2�TH2CO3��Fe2O3+6HCl=2FeCl3+3H2O��
��2��Ca��OH��2+CO2=CaCO3��+H2O��CaCO3+2HCl�TCaCl2+H2O+CO2����
��3��C3H8+5O2
3CO2+4H2O��
��2��ʯ��ˮ�е��������������ƣ���������еĶ�����̼��Ӧ����̼��ƺ�ˮ��̼�����һ�ֲ�����ˮ�İ�ɫ���ʣ���ƿ�г��γ�һ���Ĥ����Ӧ�Ļ�ѧ����ʽΪ��CO2+Ca��OH��2�TCaCO3��+H2O��̼�������ϡ���ᷴӦ�����Ȼ��ơ�ˮ�Ͷ�����̼����Ӧ�Ļ�ѧ����ʽΪ��CaCO3+2HCl�TCaCl2+H2O+CO2����
��3����֪A��һ�������к���3��̼ԭ�ӣ���A����Է�������Ϊ44�����������غ㶨�ɣ�A��һ�������к���8����ԭ�ӣ�B���ӵĻ�ѧʽΪ��O2��C�Ļ�ѧʽΪ��CO2��D�Ļ�ѧʽΪ��H2O����Ӧ�Ļ�ѧ����ʽΪ��Fe2O3+6HCl=2FeCl3+3H2O��
�ʴ�Ϊ����1��H2O+CO2�TH2CO3��Fe2O3+6HCl=2FeCl3+3H2O��
��2��Ca��OH��2+CO2=CaCO3��+H2O��CaCO3+2HCl�TCaCl2+H2O+CO2����
��3��C3H8+5O2
| ||
�����������ѶȲ�����ѧ�����ݷ�Ӧԭ����д��ѧ����ʽ����������ѧ����ʽ��д�������ֵĴ����в����Ͽ���ʵ�������������غ㶨�ɡ���д������������ŵȣ�

��ϰ��ϵ�д�
 ��Ԫ����ĩ��ϰ�ȷ��ϵ�д�
��Ԫ����ĩ��ϰ�ȷ��ϵ�д� ����ͬ�����Ծ�ϵ�д�
����ͬ�����Ծ�ϵ�д�
�����Ŀ
ȼ������ᷢչ������������Ҫ�����ã�
��1������ȼ�ϵı仯���������仯���� ��
A���ƾ��Ļӷ� B��ú��ȼ�� C�� ʯ������Һ��
��2���ƾ���Ϊȼ�ϵ�һ��������������Ҳ���Ź㷺Ӧ�ã��磺ͨ���ƾ��������ʾ����ɫ�仯�������ɿ��ٲ��˾���Ƿ�ƺ�ݳ�����Ӧԭ����2CrO3����ɫ��+3C2H5OH+3H2SO4�TCr2��SO4��3����ɫ��+3CH3CHO+6X��X�Ļ�ѧʽΪ ��
��3����������Ϊ��������ȼ�ϡ������û�ѧ����ʽ���͡�������������ԭ�� ��
��4����ͥȼ����ˮ���ϳ���ע��ȼ����ˮ������װ���ܱ�ԡ�һ�ͨ�粻�����������ͱ�ע�˱�ʶ��ԭ�� ��
��5��������Ȼ���֡�ֽ���������ͼ������ֽ���������ֽ�������������������ʢ�����ϣ�
���ƾ�ȼ��ʱֽ���ᱻ��ȼ�����ڴ��������н��Ͳ��������� ������ţ���
A�����������㣬ֽ����ȼ�� B��ֽ��ˮ��ʪ��ֽ���Ż�㽵��
C��ˮ����ʱ�����������¶ȴﲻ��ֽ���Ż�� D���ƾ�ȼ��ʱ���������٣��¶ȴﲻ��ֽ���Ż��
��6���±��г�����ÿ������ȼ����ȫȼ��ʱ�ų�����������Ԫ�ص�����������
�����������ݷ��������ѡ�õ�����ȼ���� ��ѡ������ԭ���� �� ��
��1������ȼ�ϵı仯���������仯����
A���ƾ��Ļӷ� B��ú��ȼ�� C�� ʯ������Һ��
��2���ƾ���Ϊȼ�ϵ�һ��������������Ҳ���Ź㷺Ӧ�ã��磺ͨ���ƾ��������ʾ����ɫ�仯�������ɿ��ٲ��˾���Ƿ�ƺ�ݳ�����Ӧԭ����2CrO3����ɫ��+3C2H5OH+3H2SO4�TCr2��SO4��3����ɫ��+3CH3CHO+6X��X�Ļ�ѧʽΪ
��3����������Ϊ��������ȼ�ϡ������û�ѧ����ʽ���͡�������������ԭ��
��4����ͥȼ����ˮ���ϳ���ע��ȼ����ˮ������װ���ܱ�ԡ�һ�ͨ�粻�����������ͱ�ע�˱�ʶ��ԭ��
��5��������Ȼ���֡�ֽ���������ͼ������ֽ���������ֽ�������������������ʢ�����ϣ�

���ƾ�ȼ��ʱֽ���ᱻ��ȼ�����ڴ��������н��Ͳ���������
A�����������㣬ֽ����ȼ�� B��ֽ��ˮ��ʪ��ֽ���Ż�㽵��
C��ˮ����ʱ�����������¶ȴﲻ��ֽ���Ż�� D���ƾ�ȼ��ʱ���������٣��¶ȴﲻ��ֽ���Ż��
��6���±��г�����ÿ������ȼ����ȫȼ��ʱ�ų�����������Ԫ�ص�����������
| ȼ������ | ���� | ���� | ���� | ��ϩ | ��Ȳ |
| ��ѧʽ | CH4 | C2H6 | C3H8 | C2H4 | C2H2 |
| ��������104J/g�� | 5.56 | 5.20 | 5.05 | 5.04 | 5.00 |
| ��Ԫ�ص�����������%�� | 25.0 | 20.0 | 18.2 | 14.3 | 7.7 |
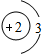 B��
B��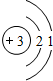 C��
C��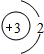 D��
D��
 22���ճ������ũҵ�����е��������ⶼ�뻯ѧ֪ʶ������أ�
22���ճ������ũҵ�����е��������ⶼ�뻯ѧ֪ʶ������أ�