��Ŀ����
����Ŀ�����Ա�ʵ�顱�ǻ�ѧѧϰ����֮��Ч��˼ά������ij��ѧѧϰС���ͬѧ��ѧ����صĻ�ѧ֪ʶ���߽�ʵ������������ʵ�飬������벢�ش��������⡣
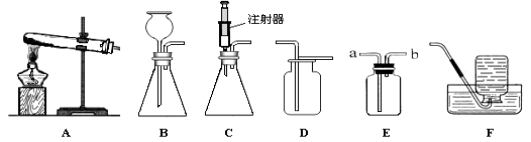
A��ȼ�յ����� B��������������� C��֤��CO2��NaOH��Ӧ D�������˶�������
��1��ͨ��ʵ��A������˵��ȼ�յ�����֮һ�� ��
��2������ʵ��B��һ��ʱ���۲쵽�Թ����е�����������ʴ���ɴ˵ó������������Ҫ����������ˮ�� ֱ�ӽӴ�������ȥ������� ϴ�ķ�����
��3��ʵ��C�����������ͬ������CO2��������ƿ��������ˮ��ƿ������NaOH��Һ��ƿ��������ʵ�飬��������ƿ���ij̶�֤��CO2 ��NaOH��Һ�е�����ȷʵ�����˷�Ӧ����һ��Ӧ�Ļ�ѧ����ʽΪ ��
��4��ʵ��D���ձ����г��ֵ�������˵�������Dz����˶��ġ����ձ�����Һ����Ũ��ˮʱ�ձ����еķ�̪��Һ����ɫ��Ϊ ɫ�����ձ�����Һ�廻��Ũ���ᣬ���ձ�����Һ�廻�ɵ��з�̪��NaOH��Һʱ��һ��ʱ�����Һ��ɫ�ı仯�� ��
���𰸡���1�����ȴﵽ��ȼ���Ż��
��2��������ϡ����
��3��CO2��2NaOH��Na2CO3��H2O
��4����ɫ��ɫ��Ϊ��ɫ��
��������
���������������ѧ֪ʶ��������Ϣ֪����1��ͨ��ʵ��A������˵��ȼ�յ�����֮һ���¶ȴﵽ��ȼ���Ż�㡣��2������ʵ��B��һ��ʱ���۲쵽�Թ����е�����������ʴ���ɴ˵ó������������Ҫ����������ˮ�Ϳ���ֱ�ӽӴ�������ȥ�������ϡ����ϴ�ķ�������3��ʵ��C�����������ͬ������CO2��������ƿ��������ˮ��ƿ������NaOH��Һ��ƿ��������ʵ�飬��������ƿ���ij̶�֤��CO2 ��NaOH��Һ�е�����ȷʵ�����˷�Ӧ����һ��Ӧ�Ļ�ѧ����ʽΪCO2��2NaOH��Na2CO3��H2O��4��ʵ��D���ձ����г��ֵ�������˵�������Dz����˶��ġ����ձ�����Һ����Ũ��ˮʱ�ձ����еķ�̪��Һ����ɫ��Ϊ��ɫ�����ձ�����Һ�廻��Ũ���ᣬ���ձ�����Һ�廻�ɵ��з�̪��NaOH��Һʱ��һ��ʱ�����Һ��ɫ�ı仯����ɫ��Ϊ��ɫ��
����������Ա�ʵ����˼ά������

����Ŀ���±���Ca��OH��2��NaOH���ܽ�����ݣ���ش��������⣺
�¶�/�� | 0 | 20 | 40 | 60 | 80 | 100 | |
�ܽ��/g | Ca��OH��2 | 0.19 | 0.17 | 0.14 | 0.12 | 0.09 | 0.08 |
NaOH | 31 | 91 | 111 | 129 | 313 | 336 | |
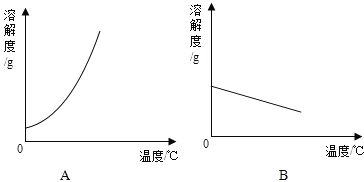
��1�������ϱ����ݣ�����Ca��OH��2��NaOH���ܽ�����ߣ���ͼ��ʾ�ܱ�ʾNaOH�ܽ�����ߵ��� _________ ����A��B����
��2��Ҫ���һƿ�ӽ����͵�Ca��OH��2��Һ��ɱ�����Һ�������ʩ�У��������������ƣ��������¶ȣ��������¶ȣ�������ˮ��������ˮ���ٻָ���ԭ�¶ȣ���������ʯ�ң�
���д�ʩ��ȷ���� _________��
A���ڢܢ� B���ۢ� C���٢ۢݢ� D���٢ڢݢ�
��3��20��ʱ��191g����NaOH��Һ������10gˮ���ٽ��µ�20����������NaOH���������Ϊ _________ ��
��4������20��ʱCa��OH��2�ı�����Һ������Һ���������м���һ����CaO��õ�����Һ������Һ������ʱ��Һ�����ʵ��������� �� _________ �ף����������������=������
��5������60��ʱ��Ca��OH��2��NaOH�������ʵı�����Һ����Ҫ�õ��ϴ�����NaOH���壬Ӧ��ȡ������������ _________ ��
��6��20��ʱ�����ⶨNaOH��Һ��pH�����Ƚ�pH��ֽ������ˮ��ʪ���ٽ��вⶨ����������Һ��pH _________ ���ƫ����ƫС������Ӱ�족����
����Ŀ����������ʵ������Ľ��ʹ������
ѡ�� | ��ʵ������ | ���� |
A | ���¹�ʮ��Ʈ�� | ���������ڲ����˶��� |
B | ˮ���¶ȼ����ˮ���������� | ԭ�Ӵ�С���¶ȸı���ı� |
C | 25m3ʯ�����ɼ�ѹװ��0.024m3�ĸ�ƿ�� | ���Ӽ����м���� |
D | һ��ˮ�к�ǧǧ�����ˮ���� | ���Ӻ�С |
A. A B. B C. C D. D
����Ŀ���±���������һЩ���ʵ�pH���й����ǵ�˵������ȷ������ ��
�������� | �������� | ����֭ | ţ�� | ��ľ��ˮ | �������� |
pH | 1 | 3 | 6.5 | 11 | 12.5 |
A. ��ľ��ˮ������ B. ���������Լ���
C. ����֭�����Ա�ţ���� D. θ�������˲��˶�Խ���