��Ŀ����
��2010?���ݣ���������������Ԫ�أ�����Ѫ�쵰�ijɷ֣��ܰ������������䣮ȱ����Ԫ�أ���ȱ����ƶѪ��ҽѧ�Ͼ�����������������Ƭ�����ֲ��˲�������ȡ20������Ƭ������Ϊ10g������40gˮ�У���������Һ�еμ��Ȼ�����Һ���Թ������ˡ�ϴ�ӡ����������֪����������Ϊ2.33g����֪��FeSO4+BaCl2=BaSO4��+FeCl2���Լ��㣺��1��������Ƭ����������������������
��2��1������Ƭ�к���Ԫ�ص�������______g��
���𰸡����������ݻ�ѧ�Ļ�ѧ����ʽFeSO4+BaCl2=BaSO4��+FeCl2�������ɵij������ᱵ����������μӷ�Ӧ������������������10g����Ƭ����������������������
���������������������������������Ƭ��������������������= ×100%����������������������ļ��㣻
×100%����������������������ļ��㣻
����������������������������������Ԫ�ص�������������Ԫ�������Ķ�ʮ��֮һ��1������Ƭ�к���Ԫ�ص�������
����⣺��1����μӷ�Ӧ����������������Ϊx
FeSO4+BaCl2=BaSO4��+FeCl2
152 233
x 2.33g
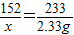 x=1.52g
x=1.52g
������Ƭ��������������������=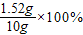 =15.2%
=15.2%
�𣺸�����Ƭ��������������������Ϊ15.2%��
��2��1������Ƭ�к���Ԫ�ص�����=��1.52g×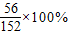 ��×
��×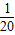 =0.028g
=0.028g
�ʴ�Ϊ��0.028��
���������ݻ�ѧ����ʽ��ʾ��Ӧ�����ʵ������ȣ����ɷ�Ӧ��ijһ���ʵ��������㷴Ӧ���������ʵ�������
���������������������������������Ƭ��������������������=
 ×100%����������������������ļ��㣻
×100%����������������������ļ��㣻����������������������������������Ԫ�ص�������������Ԫ�������Ķ�ʮ��֮һ��1������Ƭ�к���Ԫ�ص�������
����⣺��1����μӷ�Ӧ����������������Ϊx
FeSO4+BaCl2=BaSO4��+FeCl2
152 233
x 2.33g
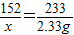 x=1.52g
x=1.52g������Ƭ��������������������=
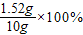 =15.2%
=15.2%�𣺸�����Ƭ��������������������Ϊ15.2%��
��2��1������Ƭ�к���Ԫ�ص�����=��1.52g×
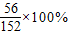 ��×
��×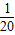 =0.028g
=0.028g�ʴ�Ϊ��0.028��
���������ݻ�ѧ����ʽ��ʾ��Ӧ�����ʵ������ȣ����ɷ�Ӧ��ijһ���ʵ��������㷴Ӧ���������ʵ�������

��ϰ��ϵ�д�
 ���ĺ����Ͼ�������ϵ�д�
���ĺ����Ͼ�������ϵ�д�
�����Ŀ
 CO2��ֻ��һ�������弴�ɣ�______��
CO2��ֻ��һ�������弴�ɣ�______��