��Ŀ����
��Դ�����ϡ�������ʳƷ������Խ��Խ�������ǵ����ӡ���ӻ�ѧ���ӽǷ��������й����Ⲣ��д�ո�
(1)��ʯȼ�ϰ���ú��ʯ�ͺ�_______________��
(2)���Ͻ����������ɻ�����ǣ����Ͻ�����_______(��ϳɲ��ϡ����������ϡ����ϲ��ϡ�)��
(3)������Ϊ���ǵ�����������ͬʱ��Ҳ�Ի�����ɲ���Ӱ�졣���ǰ����ϸ�����������Σ����Ϊ��____________����
(4)Ϊ�˺�����ʳ��Ӫ�����⣬�����а�ե�õ��߲�֭�ŵ�����У����ɿ�ζ���ء�Ӫ���ḻ���߲���ͷ��������ͷ�ṩ����ҪӪ��������_____��___________��
(5)���ʶ����ũ�������������Ҫ���á�����[CO(NH2)2]����_____(����ʡ����طʡ����ʡ�)��
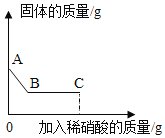
ʵ��������һƿ���������Ȼ��ƺͿ��������ʵ���������Ʒ��ij��ѧ��ȤС��Ը���Ʒ�ɷֽ��ж����ⶨ���ס��ҡ�����λͬѧ�ֱ��ȡһ����������Ʒ�����ձ��У���������ˮ����˵õ���Һ��������Һ�м���һ���������Ȼ�����Һ����ַ�Ӧ����ˣ�������ϴ�ӡ����ﲢ���������ʵ�����ݼ�¼���£�
������Ŀ | �� | �� | �� |
��ȡ��Ʒ������/g | 15 | 15 | 20 |
�����Ȼ�����Һ������/g | 150 | 100 | 100 |
��Ӧ�����ó���������/g | 23.3 | 23.3 | 23.3 |
��ش��������⣺
��1�� ͬѧ��ѡ��ס����ҡ���������ȡ�õ���Ʒ���Ȼ�����Һǡ����ȫ��Ӧ��
��2���������Ʒ�������Ƶ�������������д�����㲽�裬������������0.1%��
��3�������������Ʒ���Ȼ�����Һǡ����ȫ��Ӧʱ������Һ��������������������ʵ������л���Ҫ�ⶨ�������� ��
�����й�ʵ����Ӻ������ǣ�������
ѡ�� | ���ʣ�������Ϊ���ʣ� | �Լ��� |
A | C��Fe�� | ��ȼ |
B | CO2��H2O�� | Ũ���� |
C | NaOH��Na2SO4�� | �����������Ȼ�����Һ�����ٲ������� |
D | BaSO4��BaCO3�� | ������������Һ������ |
A.A B.B C.C D.D


 ��
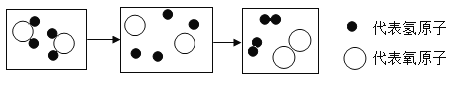
��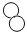 ���Ӹ�����Ϊ1:2
���Ӹ�����Ϊ1:2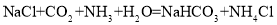 ���������ġ������Ƽ������Ҫ��Ӧ�������й�������ȷ����( )
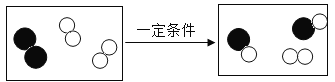
���������ġ������Ƽ������Ҫ��Ӧ�������й�������ȷ����( )