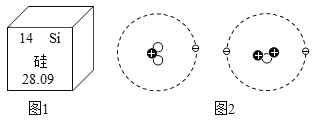
��Ŀ����
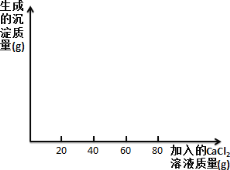
ijʳ�ü����Ҫ�ɷ��� Na2CO3�����к��������� NaCl��С��ͬѧΪ�ⶨ��ʳ�ü��� Na2CO3 ������������������������ʵ�飬ȡ 40 g ʳ�ü��ˮ��� 400g ����Һ������Һƽ����Ϊ�ķݣ�Ȼ��ֱ����һ������������ CaCl2 ��Һ��ʵ�����ݼ��±���
ʵ��һ | ʵ��� | ʵ���� | ʵ���� | |
ʳ�ü������/g | 10 | 10 | 10 | 10 |
����CaC12��Һ������/g | 20 | 40 | 60 | 80 |
���ɳ���������/g | 2.5 | 5 | m | 8 |
������������ݲ�����ش�
��1��m��________��
��2��40 g ��ʳ�ü���ɵ���Һ������ CaCl2 ��Һ��Ӧ��������ɳ�������Ϊ_____g��
��3����ʳ�ü��� Na2CO3 �����������Ƕ���_____����д��������̣��𰸾�ȷ�� 0.1%��
��4�������ʵ���ĵ����ݻ�ͼ�����Ϻ��ʵ���������_____��
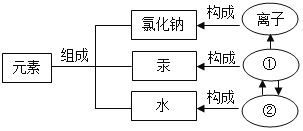
���и��ݻ�ѧ˼ά�ó��Ľ�����ȷ����
ѡ�� | ���� | ʾ�� |
A | �غ㷨 | 2 ��þ��������ȼ�����ù�������Ϊ 2 �� |
B | �Աȷ� | �ҳ������������ֻ��Ҫ�������Ա�ʵ�� |
C | ������ | ������ֻ��һ��Ԫ�أ�����ֻ��һ��Ԫ�ص����ʾ��ǵ��� |
D | ���ɷ� | ���ɱ������ɷ��ӹ��ɵ� |
A.A B.B C.C D.D
��ͼ��ʾ�������������˵����ʣ���һ���������¶��ܷ�����ѧ��Ӧ����������ǣ�������

�� | �� | �� | �� | |
A | C | O2 | H2SO4 | CuO |
B | Fe | H2SO4 | CuCl2 | NaOH |
C | NaOH | CO2 | KOH | CuCl2 |
D | Na2SO4 | Ba(OH)2 | HCl | Ba(NO3)2 |
A.A B.B C.C D.D



 B
B