��Ŀ����
�����ѧϰ��������ἴ�̲ġ�����ѧ����ʳס�С�������Դ�����Ͽ�ѧ��ҽ�������ȷ���Խ��Խ��������Լ��ļ�ֵ��
��1��һ������ҩ������������ͭ��ȼ��ʱ������ɫ���棬��ѧ����ʽ���£�
2Cu��NO3��2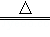 2CuO+O2+4X������X��һ�ֿ�����Ⱦ��仯ѧʽΪ ��
2CuO+O2+4X������X��һ�ֿ�����Ⱦ��仯ѧʽΪ ��
��2��������ԭ����һ����̼����������Ӧ��д���䷴Ӧ�Ļ�ѧ����ʽΪ ��
��3��С����̽�����ƽ����������Ƿ��н���ͭ������ѡ�������Լ��е� ��
A��ZnC12��Һ B��Mg��NO3��2��Һ C��H2SO4��Һ D��AgNO3��Һ
��4��Ϊ̽��������ȼ�յ���������ͬʱ��ȼСľ����Сú�飬����Сú���ȼ����ʱ�䳤��ԭ���� ��
��5���������������pHΪ6���ҵ������У���ij������pHΪ4�������������ֲ��ȣ����������п����������������Ե��� ��
A����ʯ�� B����ʯ�� C��������� D���������ƣ�
��1��NO2����2��3CO+Fe2O3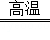 2Fe+3CO2����3��D����4��B��
2Fe+3CO2����3��D����4��B��
���������������1���ɷ�Ӧ�Ļ�ѧ����ʽ2Cu��NO3��2�T2CuO+O2��+4X�����ɵ�֪��Ӧǰ���ԭ�ӵĸ���Ϊ��
��Ӧǰ��Ӧ��
ͭԭ�� 2 2
��ԭ�� 4 0
��ԭ�� 12 4
���ݻ�ѧ�仯ǰ��ԭ�ӵ����ࡢ��Ŀ���䣬������X��4�������к���4��Nԭ�Ӻ�8��Oԭ�ӣ���ÿ��X������1��Nԭ�Ӻ�2��Oԭ�ӹ��ɣ�����X�Ļ�ѧʽΪNO2��
��2��һ����̼����������Ӧ���ɶ�����̼����������ʽΪ��3CO+Fe2O3 2Fe+3CO2��
2Fe+3CO2��
��3���������ǿ�Ľ����ܹ��ѽ���������Ľ�����������Һ���û�������������п��þ�Ļ�Ա�ͭǿ������ͭ�������Ȼ�п������þ��Ӧ����˲���ȡ��ͭ�Ļ�������������ͭҲ������ϡ���ᷴӦ�����Ҳ����ȡ��ͭ�Ľ�����Ա���ǿ������ͭ���û����������еĽ�������˿��������жϡ��ƽ����������Ƿ��н���ͭ��
��4�����pHԽ��������Խ����������Ҫ����ij������pHΪ4��Ϊ6�ķ����Ǽ����Լ��Ե����ʶ���������ģ�A����ʯ������ˮ�����ɼ��Ե��������ƣ����ù��̻�ų��������ȣ������������������ʧ�����Բ���ȡ��B�����������Լ��ԣ��ܹ��к����Ե�������������Ŀ��Ҫ��C��������Ƶijɷ����������ƺ�����ƻ��������ԣ��������������������ԣ�D�����������Լ��ԣ��ܹ������ᵫ�����̫ǿ����˲������ڸ���������������ѡB
���㣺�����غ㶨�ɼ���Ӧ�ã������Ļ�ѧ���ʣ��кͷ�Ӧ����Ӧ�ã���д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ��ȼ����ȼ�յ�����

 ���ٴ���������ѧϰ����ѧ�ں����ν�ϵ�д�
���ٴ���������ѧϰ����ѧ�ں����ν�ϵ�д�